Scientists produce polyamide membranes with nanoscale Turing structures
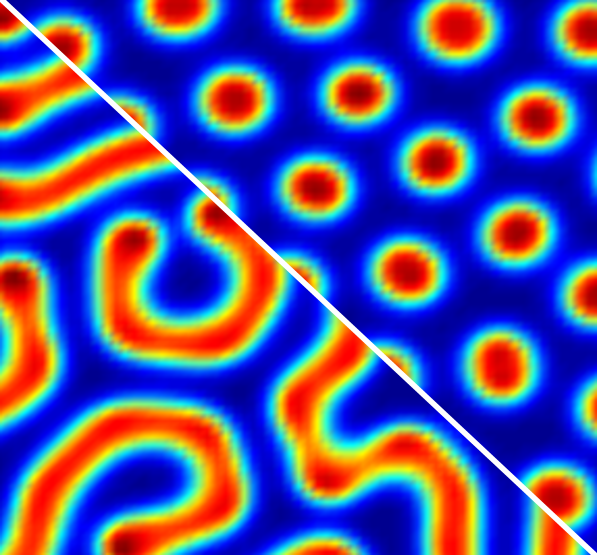
The zebra’s stripes, the snail’s spirals and the leaf’s convolution……It has been an intriguing enigma as to how these regular and repetitive patterns form in nature. As early as 1952, English scientist Alan Turing made a prediction that two chemical substances, activator and inhibitor, can, under certain conditions, react and diffuse with each other to generate spatiotemporal stationary structures. Through what is dubbed as a “reaction-diffusion” process, these two substances will spontaneously form such patterns as stripes, spots or spirals. This hypothesis has been corroborated by succeeding scientists and this pattern is termed as the “Turing” structure.
Recently, a research team led by Prof. ZHANG Lin, a professor of biomass chemical engineering who is dedicated to research into membranes, integrated Turing structures with membrane studies and pioneered in creating polyamide membranes with nanoscale Turing structures. Their findings are published in the May 4 issue of the journal of Science.
TAN Zhe, a Ph.D. candidate in the College of Chemical and Biological Engineering, is the leading author and Prof. ZHANG Lin is the corresponding author. Participants in this research also included Prof. CHEN Shengfu and Prof. GAO Congjie with the College of Chemical and Biological Engineering and Prof. PENG Xinsheng with the School of Materials Science and Engineering.

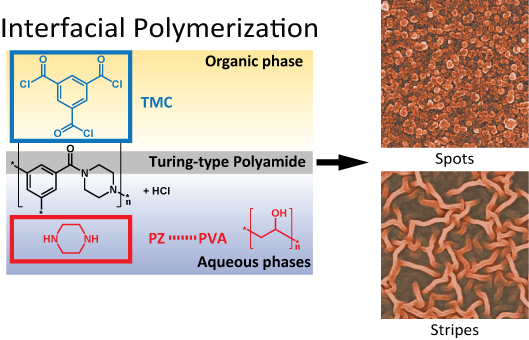
Interfacial polymerization, as a common technique, has been applied to prepare super-thin membranes. However, despite the fact that preparing reverse osmosis and nanofiltration membranes is identical in terms of technology and reaction mechanism, there exist remarkable distinctions in surface structure. In texture, nanofiltration membranes are very silky whereas reverse osmosis membranes are relatively coarse.
Why do they differ so enormously from each other? There is no definite and conclusive answer.
Prof. ZHANG Lin et al. decided to conduct in-depth research into this “overlooked” question. While delving into the underlying reason, they found out that the interfacial polymerization process is a typical “reaction-diffusion” system. Much to their excitement, they associated it with the conditions for Turing structures.
Turing structures arise when imbalances in diffusion rates make a stable steady-state system sensitive to small heterogeneous perturbations. For example, Turing patterns occur in chemical reactions when a fast-moving inhibitor controls the motion of a slower-moving activator.
Researchers used a facile route based on interfacial polymerization to grow polyamide membranes, where the reactions occur at the interface between oil and water layers. The addition of polyvinyl alcohol to the aqueous phase reduced the diffusion of the monomer. This process generated membranes with more bumps, voids, and islands, which prove to be better for water desalination.
This work demonstrates that Turing structures can be produced by interfacial polymerization when appropriate initial conditions are created. Microscopic characterization of the Turing-type membranes reveals that the spatial distribution of relatively higher water permeability sites agrees well with the corresponding Turing structures at the nanoscale. These unusual nanostructures, which are generated by diffusion-driven instability, enable outstanding transport properties in both water permeability and water-salt selectivity.