ZJU scientists reveal key regulator for cell cycle
The research team headed by WANG Fangwei, a professor with Life Sciences Institute and Innovation Center for Cell Signaling Network at Zhejiang University, published an article entitled “WAC Promotes Polo-like Kinase 1 Activation for Timely Mitotic Entry” in the July 17 issue of the journal of Cell Reports.
Cell division is the process by which a parent cell divides into two or more daughter cells. It usually occurs as part of a larger cell cycle. Actively dividing eukaryote cells pass through a series of stages known collectively as the cell cycle: two gap phases (G1 and G2); an S (for synthesis) phase, in which the genetic material is duplicated; and an M phase, in which mitosis partitions the genetic material and the cell divides.
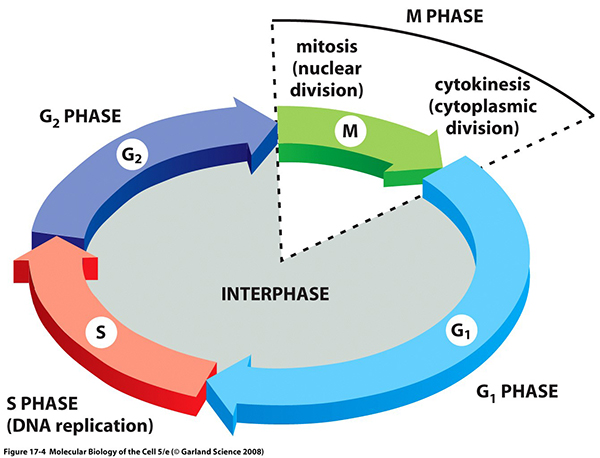
Polo-like kinase 1 (Plk1) plays a key role in many steps of mitotic cell division, ranging from mitotic entry to cytokinesis. During G2 phase of human cells, Aurora A kinase (AurkA) cooperates with its co-factor Bora to phosphorylate Plk1 at Thr-210 in its activation loop, which is important for Plk1 activation and the timely entry into mitosis. Several lines of evidence suggest that Plk1 phosphorylation at T210 (Plk1-pT210) occurs at the centrosome, which is consistent with the cytoplasmic localization of Bora in interphase cells. However, Plk1 activity is first seen in the nucleus of early G2 cells. The mechanism underlying Plk1 activation during the G2 to mitosis (G2/M in short) transition remains far from fully understood.
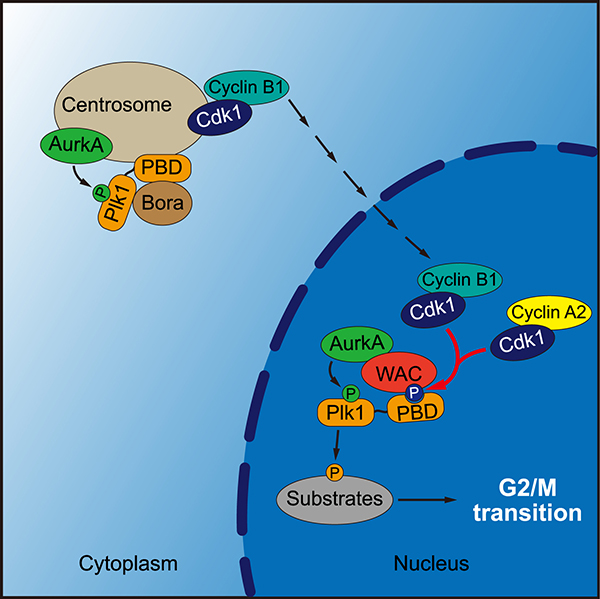
WANG et al. report that the activation of Plk1 requires WAC, a WW domain-containing adaptor protein with a coiled-coil region that predominantly localizes to the nucleus in interphase. Cyclin-dependent kinase 1 (Cdk1) phosphorylates WAC, priming its direct interaction with the polo-box domain of Plk1. Knockdown of WAC compromises Plk1 activity and delays mitotic entry. These defects are rescued by exogenous expression of wild-type WAC, but not the Plk1-binding-deficient mutant. WAC also binds AurkA and can enhance Plk1 phosphorylation by AurkA in vitro.
Taken together, these results indicate an indispensable role for WAC in promoting Plk1 activation and the timely entry into mitosis. Thus, WAC is a versatile adaptor protein involved in multiple cellular processes under various cellular contexts.