The “black box” behind gas molecules reacting at catalytic sites unlocked
Why has the catalytic reaction remained enigmatic for so long? The scientific community has failed to directly visualize gas molecules reacting at catalytic sites even with immense and persistent efforts in the past. It can be for the main part ascribed to the fact that the molecules that adsorb and react dynamically do not offer sufficient contrasts for transmission electron microscopy (TEM) identification. ZHANG Ze, CAS Fellow from the State Key Laboratory of Silicon Materials and Center of Electron Microscopy in the Zhejiang University School of Materials Science and Engineering, drew an apt analogy between tiny dusts in the air and gas molecules. Because of their low contrast (weak scattered light) in the air, it is difficult for human eyes to identify dusts.
So how can gas molecules at catalytic sites be observed? One day, when WANG Yong, a professor from the Zhejiang University School of Materials Science and Engineering, and ZHANG Ze were discussing this issue in the office, a beam of sunlight shone through the window, and those originally invisible dusts appeared immediately. They realized out of the blue that if they wanted to visualize gas molecules with a weak contrast in the reaction, they would first find that beam of magic “sunshine”.
To this end, they chose extensively-studied titanium dioxide (TiO2), which is non-toxic and harmless to the environment, as the very catalyst to react with water. The TiO2 surface is marked by the highly ordered four-coordinated Ti (Ti4c) rows (termed “active rows,” owing to their lower coordination). This could facilitate an enhanced contrast of adsorbing molecules along the row direction and allow real-time monitoring of H2O species dissociating and reacting on the catalyst surface.
The atomic structure of the TiO2 (1×4)-(001) surface was characterized by both aberration corrected ETEM and scanning transmission electron microscopy (STEM) images. The bulk truncated (1×1)-(001) surface reconstructed to a (1×4)-(001) surface by periodically replacing the surface oxygen rows (along the [010] direction) with TiO3 ridges every four unit cells along the TiO2 [100] direction. As a result, protruded Ti4c rows were periodically exposed on the surface and provided striking contrasts, so the subtle changes occurring in reactions could be detected by means of ETEM observation without contrast overlapping. The ordered Ti4c active rows could offer sufficient contrasts for direct ETEM visualization of water if the molecules adsorbed in ordered arrays.
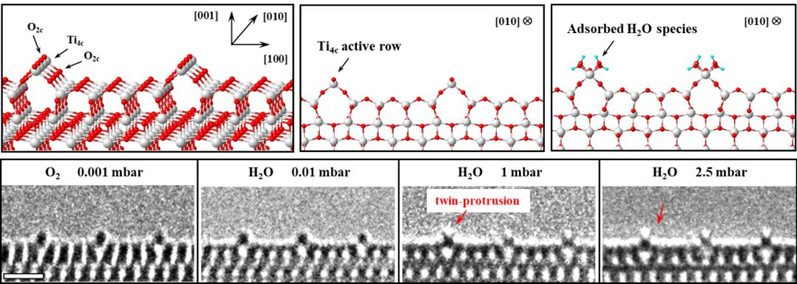
When H2O vapor was adsorbed on protruded Ti4c rows, two additional small protrusions were observed at the top of the Ti4c rows during experiments. This twin-protrusion structure, resembling two “rabbit ears”, was visible during the TEM observation. When the background environment was changed from H2O to O2 or vacuum, the twin-protrusion structure faded away.
The identification of this twin-protrusion structure was a crucial breakthrough in that the two “rabbit ears” offered valuable clues to “seeing the catalytic reaction clearly”. “It is very difficult to determine or predict these unique structural changes in the catalytic reaction by other means,” said ZHANG Ze, “Imaging at the atomic scale with TEM benefits from the development of aberration correctors. Coupled with the development of in-situ reaction technology in TEM, it is possible to observe solid samples at the atomic scale in the gas or liquid environment.”
It is the first time that the scientific community has directly monitored water molecules dissociating and reacting on the catalyst surface. Another highlight of this study is to introduce gas into an electron microscope to simulate an actual chemical reaction environment in various aspects, including temperature and pressure.
Relevant findings are published in an article entitled “Visualizing H2O molecules reacting at TiO2 active sites with transmission electron microscopy” in the January 24 issue of Science.
