A patch for activation of anticancer drug inside tumor
Pharmaceutical researchers have developed a new technique that uses a patch with microneedles to perform catalyzed reactions for generating anticancer therapeutics directly inside tumors. Early laboratory tests on mice show it potentiates the effectiveness of drugs and reduces toxicity. The technique was developed by researchers at Zhejiang University, Fuzhou University and the University of California, Los Angeles.
“This is a proof of concept, but we’ve demonstrated there is now a new tool for activating anti-cancer drugs directly inside tumor tissue – and that should make drug treatments significantly more effective, meanwhile minimize potential side effects,” said Dr. GU Zhen senior author of a paper published in Nature Nanotechnology and a professor at the Zhejiang University College of Pharmaceutical Sciences.
The researchers applied a reaction named transition metal-mediated bioorthogonal catalysis, which happens within the microneedles. “We call our device ‘Bioorthogonal Catalytic Patch’ loaded with palladium nanoparticles as catalysts. It could mediate the bioorthogonal transition and activation of a prodrug, which was initially administered to the body in an inactive (or less than fully active) form, and then can be converted to its active form with anticancer function,” said Dr. CHEN Zhaowei, the lead author of this study.
The microneedle array patch was fabricated using polyvinyl alcohol (PVA) as the matrix, and using palladium nanoparticles-deposited titanium dioxide nanosheets as doping agents. Palladium nanoparticles could catalyze the “decagging reaction” of N-(allyloxycarbonyl)-protected drug molecules (prodrugs), turning the less toxic prodrugs into drugs with anti-tumor effects. As the titanium dioxide nanosheets are rich in hydroxyl groups, they can be used as a support matrix for palladium nanoparticle catalysts and also, they can evenly distribute in the PVA matrix, giving the microneedles enough strength to penetrate the tumor in a minimally invasive manner.
Utilizing a mouse melanoma model, the prodrugs of doxorubicin (a chemotherapeutic drug), which were injected into the body through systemic administration, could be activated in situ in the tumors with pre-inserted microneedle patch. The locally generated doxorubicin then significantly inhibited tumor growth. With this bioorthogonal catalytic patch device, local activation of the prodrug can not only increase the dose of the drugs to be administered, but also reduce toxicity and side effects of the anticancer drug on healthy organs and tissues. Of note, this microneedle array patch still has enough mechanical strength, which facilitated the removal of the entire device after use, and avoids residual transition metal in the body, inflammation or other side effects.
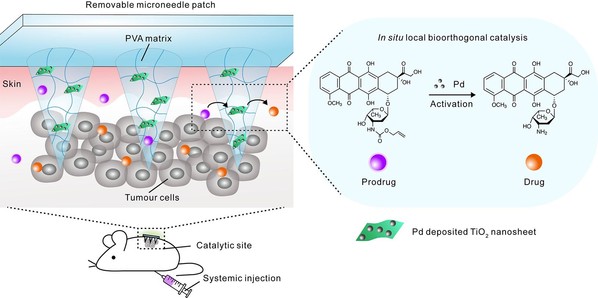
Schematic illustration of in situ bioorthogonal catalysis mediated by microneedle patch for locally activating systematically administrated prodrugs in vivo.

(a) Photograph and (b) SEM image of a Pd-TNSs incorporated microneedle array patch (PT-MNs). Scale bar in b: 500 µm. (c) SEM images showing the longitudinal section of one microneedle (top, scale bar: 200 µm) and the distribution of Pd-TNSs inside the matrix (bottom, scale bar: 20 µm).
In previous studies, Gu and colleagues have developed a blood sugar-responsive “smart insulin patch”, which proposed a new concept of bio-responsive transdermal drug delivery systems. In this work, they used bioorthogonal catalyst-loaded microneedle patch as a convenient device to achieve the local activation of anticancer prodrugs within the tumor microenvironment in a simple and effective way. Moreover, the toxic catalyst in the needle can be removed from the body together with the microneedle patch.
“We expect that the invention of this new device could further help the development of more efficient drug formulations with minimized side effects for treating other diseases,” Gu said.
Source: Zhejiang University College of Pharmaceutical Sciences