ZJU scientists unravel mechanism of genome instability by DNA Pol μ misincorporatio
Endogenous and exogenous factors may result in a variety of DNA damage, among which DNA double-strand breaks (DSBs) are the most perilous type of DNA damage. If they fail to be repaired promptly, it will affect genome stability considerably and even lead to cell death. Nonhomologous end joining (NHEJ) is one of the main pathways to repairing DSBs.X family polymerases (Pol μ, Pol λ, and TdT) are involved in the NHEJ pathway and perform gap-filling repair synthesis. Pol μ can effectively misinsert dGTP opposite 1-nt gapped DNA containing templating T. This misincorporation further facilitates subsequent NHEJ ligation reaction by the DNA ligase IV/XRCC4 complex, which may ultimately lead to genomic instability during NHEJ repair. However, the mechanism of Pol μ-induced genome instability remains obscure.
The research team led by Prof. ZHAO Ye and Prof. HUA Yuejin from the Zhejiang University College of Life Sciences published a research article entitled “Mechanism of genome instability mediated by human DNA polymerase mu misincorporation” in the journal Nature Communications on June 18. In this study, researchers investigated the misincorporation of dGTP by Pol μ, discovered key amino acid sites where nucleotides are identified and inserted, and elucidated on the molecular mechanism for genomic instability mediated by DNA polymerases.
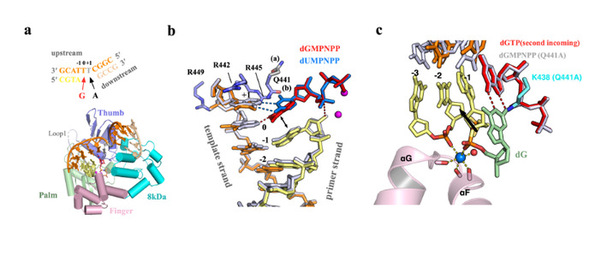
Previous studies revealed that Pol μ skips the templating base and uses the gapped DNA substrate on the downstream to maintain the canonical Watson–Crick (WC) pair, which may well bring about DNA mutations. Prof. ZHAO Ye and Prof. HUA Yuejin determined and compared crystal structures and kinetics of Pol μ substituting dGTP for dATP on gapped DNA substrates containing templating T. Pol μ is highly mutagenic on a 2-nt gapped DNA substrate, with T:dGTP base pairing at the 3ʹ end of the gap. Two residues (Lys438 and Gln441) interact with T:dGTP and fine-tune the active site microenvironments. The in-crystal misincorporation reaction of Pol μ revealed an unexpected second dGTP in the active site, suggesting its potential mutagenic role among human X family polymerases in NHEJ.
On the whole, the misincorporation of DNA polymerases may cause base substitutions or frameshift mutations and eventually genomic instability. “This study reveals different mechanisms of Pol μ’s dGTP misincorporation and implies that as compared with other polymerases, Pol μ has a potentially stronger mutation-inducing propensity in the NHEJ pathway,” said Zhao.