Novel therapeutics against obesity by activating melanocortin-4 receptor
Satiety in the gut is regulated by the nervous system. The melanocortin-4 receptor (MC4R) in the hypothalamus is known as the brain’s “switchboard” to control appetite and metabolism. It is a popular target for the treatment of such metabolic diseases as obesity by regulating the body’s energy metabolism and eating behavior in response to the simulation of proopiomelanocortin (POMC) and agouti-related peptide (AgRP) by various metabolic signals such as leptin and neuropeptide Y. MC4R belongs to the melanocortin receptor family, a group of five (MC1R−MC5R) class A G protein-coupled receptors (GPCRs) and is primarily coupled to the stimulatory G protein (Gs) and increases intracellular cyclic adenosine monophosphate (cAMP) accumulation. However, the five members of this family are highly conserved in amino acid sequence and they recognize identical or very similar peptide ligands. Owing to the exceptional sequence homology among MCRs, most peptidic agonists initially developed for MC4R lack the capability of avoiding cross-reactivity with other MCR subtypes, such as MC1R, leading to adverse effects, e.g., skin pigmentation and nausea. Thus, it is of immense necessity to address the heterogeneity of the receptor in developing drugs targeting MC4R.
The research team led by Prof. ZHANG Yan from the Zhejiang University School of Medicine and the research team led by WANG Mingwei from the Fudan University Department of Pharmacology conducted collaborative research in this field. Their research findings were published in an article titled “Structural insights into ligand recognition and activation of the melanocortin-4 receptor” in the journal Cell Research on August 25.
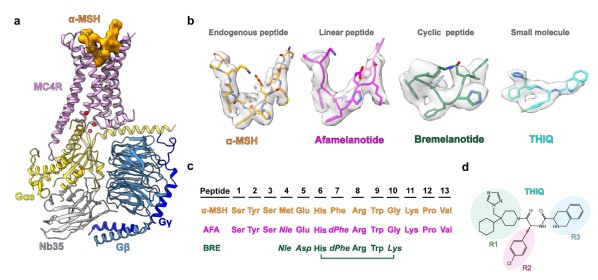
Cryo-EM structures of the MC4R–Gs complexes
Researchers employed single-particle cryo-electron microscopy (cryo-EM) to determine four structures of the human MC4R−Gs complexes: three bound to α-MSH, afamelanotide or bremelanotide at the resolutions of 3.0 Å, 3.0 Å and 3.1 Å, respectively; and one bound to THIQ at the resolution of 3.1 Å. These structures reveal the molecular basis of ligand recognition and receptor activation for MC4R, shed light on the subtype selectivity of MCRs, and provide templates for the rational design of novel therapeutics targeting MC4R to treat obesity.
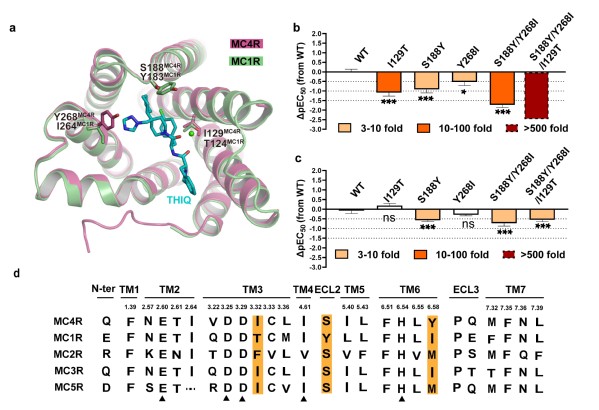
Selective activation of MC4R by THIQ
The amino acid sequence of MCRs is highly conserved. Except MC2R, the other four MCRs, which are endogenously activated by melanocyte-stimulating hormones (MSH), are virtually non-selective. Researchers made a detailed analysis of the structural basis of the selectivity of THIQ, a small-molecule agonist initially developed by Merck in 2005. By comparing the structure of MC4R and homology-modeled MC1R, they discovered that three amino acids interact differently with THIQ in the two receptors. Subsequent experiments revealed that the combination of these 3 amino acids played a vital role in THIQ selectivity. In addition, researchers also identified more concentrated regions of non-conserved amino acid aggregation that could be instrumental for the development of selective ligands.
“These results offer unprecedented structural insights into the pharmacological and signaling features of MC4R and deepen the understanding of how naturally occurring mutants of MC4R modify the receptor activity,” said Prof. Zhang. “Our research will serve as a structural template for rational drug design targeting the leptin-melanocortin pathway and facilitate the discovery of novel therapeutics against obesity.”