Metabolic remodeling during murine early embryo development
The research team led by Prof. ZHANG Jin from the Zhejiang University School of Medicine and the team led by Dr. HU Zeping from the Tsinghua University School of Pharmaceutical Sciences published their collaborative research regarding metabolic remodeling during murine early embryo development in the journal Nature Metabolism on October 14.
Component analysis of the fallopian tubal liquid and identification of suitable medium ingredients for culturing mammalian embryos in vitro highlighted the importance of studying metabolism in controlling early embryo development several decades ago. Recent studies have revealed the impact of certain metabolites and metabolic pathways on embryos of different stages or pluripotent stem cells in different states. However, comprehensive understanding of metabolic reprogramming and its influence on epigenetic regulation in early embryo development remains elusive.
To better understand it, the research team used a mass spectrometry-based ultra-sensitive targeted metabolomics approach to directly measure metabolite abundance in murine embryos of 2-cell and blastocyst stage, and in their in vitro counterparts of 2-cell like cells and ES cells representing the totipotent state and the pluripotent state, respectively. By correlating metabolomics profiling with transcriptomics analysis, researchers illustrated unique metabolic signatures of 2-cell and blastocyst stage embryos, and a transition from a more reduced state to an oxidative state, with OxPhos genes activated by upstream developmental transcription factors such as Esrrb, Myc and Klf.
Interestingly, a reciprocal relationship between α-ketoglutarate (α-KG) and the competitive inhibitor of α-KG-dependent dioxygenases L-2-hydroxyglutarate (L-2-HG) is consistent with the redox difference between blastocyst and 2-cell embryos. High L-2-HG, but not its enantiomer D-2-HG, in the 2-cell embryos inherited from oocytes and 1-cell zygotes (in molar range) markedly decreased during the embryo development, with α-KG picking up in the opposite direction. Treating embryos with a permeable octyl-L-2-HG or knocking down L2hgdh, a gene encoding the L-2-HG consuming enzyme, resulted in delayed histone methylation erasure and embryo morphological abnormalities, suggesting that the decrease of L-2-HG is essential for global histone methylation remodeling during preimplantation development.

L-2-HG, the enantiomer of D-2-HG, was found to be present in the mouse oocytes and early embryos, and involved in the epigenetic remodeling of early embryo development.
Meanwhile, this research provided comprehensive insights of dynamic and interconnected metabolic, transcriptional and epigenetic network remodeling during murine early embryo development.
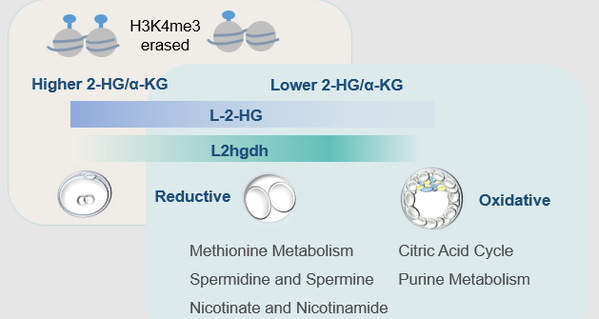
A summary of metabolic remodeling during early mouse embryo development
“Our research will enrich the knowledge of early embryo development and be valuable for optimizing embryo culture in vitro,” said Prof. Zhang.