New insights into asthma drug development
Asthma is one of the most common chronic respiratory diseases worldwide and its prevalence is on the rise. Epidemiological studies reveal that the incidence of asthma ranges from 1% to 18% in various countries. More than 400 million people are suffering from asthma all over the world. The number of asthma patients aged 20 years and above in China has added up to 45.7 million. However, the treatment of asthma is far from satisfactory. Therefore, it is of urgent need to explore the pathogenesis of asthma and identify new therapeutic targets.
Prof. SHEN Huahao and Prof. YING Songmin at the Second Affiliated Hospital of the Zhejiang University School of Medicine teamed up with Prof. ZHANG Yan at the Zhejiang University School of Medicine to identify and characterize different N-terminal truncations of endogenous chemokine CCL15 as balanced or biased agonists targeting CCR1, laying a solid foundation for the development of asthma drugs targeting CCR1. Their findings were published in the journal Nature Chemical Biology on December 24.
As early as 2003, SHEN Huahao pioneered in demonstrating that eosinophils could directly induce asthma. With unremitting efforts in more than 10 years, Shen and Ying further elucidated the key role of chemokine receptor CCR1 activation. They identified CCR1 and its ligands as new targets for asthma treatment for the first time.
Nevertheless, for nearly two decades, all small molecule drugs for asthma targeting chemokine receptors have failed in Phase II clinical trials.
“We have clearly identified the vital key of CCR1 activation in inflammation, but we cannot figure out a feasible way to inhibit it. It is because we fail to decipher its intrinsic mechanism. Because of that we cannot precisely regulate it,” said Ying, “We hope that we can gain a perspicacious understanding of the interaction between CCR1 and its ligands so as to unravel this enigma.”
Chemokine receptor CCR1 is one of the types of G protein-coupled receptor (GPCR). GPCRs constitute the largest family of transmembrane proteins and are the most important drug target in the human body. Prof. ZHANG Yan has been long committed to study in GPCR signaling and structural pharmacology. He has developed cryogenic-electron microscopy-based GPCR structural pharmacology and has made a series of achievements in the field of GPCR structural pharmacology.
Thus, Shen, Ying and Zhang made concerted efforts to resolve the high-resolution structure of the G protein complex of CCR1 bound to different CCL15 truncations.
They found that the increasing secretion of metalloenzymes in inflammation acts like a sharp knife which can cut CCL15 ligands into three subtypes with different lengths: CCL15L, CCL15M, and CCL15S. These three chemokines can lead to different signaling pathways.
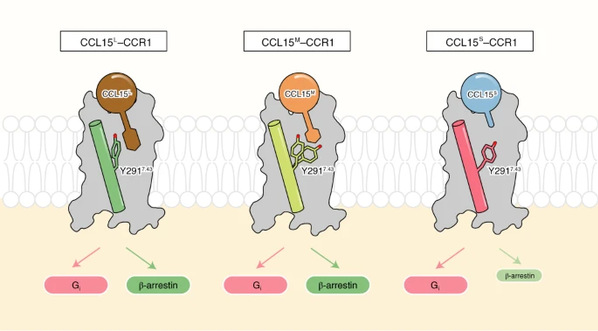
Cryo-EM density maps revealed that the side chain of Tyr291 (Y2917.43) in the orthosteric binding pocket of CCR1 exhibited remarkably distinct conformations with different ligands. The biased signaling properties of CCL15 were virtually vanishing into the air after Y2917.43 was mutated, suggesting that Y2917.43 is the key amino acid site mediating the biased activation of CCL15 on CCR1. Together with pharmacological and computational studies, this study revealed the indispensable role of Y2917.43 in identifying GPCR ligands and signaling.
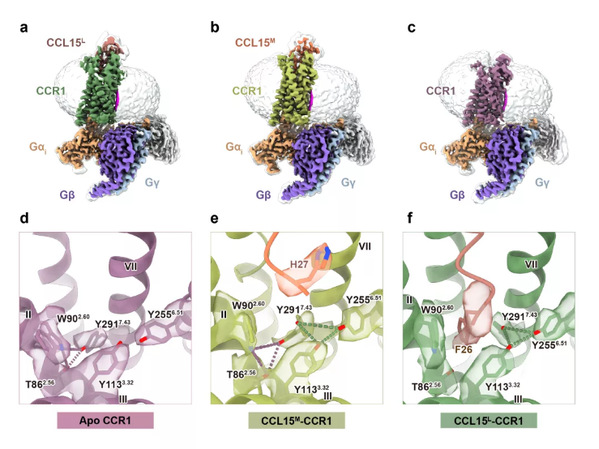
Cryo-EM structures of CCR1–Gi complexes
“Y2917.43 is like a railroad switchman that determines the direction of signals,” said Zhang. “Once we know how signaling molecules activate CCR1, we will be able to work out ways to hinder them from working. As a consequence, we may consider how to seal this ‘switchman’ in our future work.”
“The ultimate purpose of basic research is to serve clinical medicine. Next, we will work on the development of structure-oriented small-molecule drugs targeting CCR1. So far, we have made some progress in research into small-molecule drugs and the identification mechanism of CCR1,” said Shen.