Macrophage-evading and tumor-specific apoptosis inducing nanoparticles for targeted cancer therapy
In the past decade, nanodrugs have shown considerable advances for cancer therapy. However, despite the successful application of several nano-formulations, most nanodrugs failed for clinical translation due to their insufficient tumor accumulation, and consequently, compromised anticancer efficacy. MPS, which formed by resident macrophages in the liver and spleen, has been demonstrated to sequester the majority of administered nanoparticles, leading to impeded delivery of therapeutics to the tumor site and even systemic toxicity. Poly lactic-co-glycolic acid (PLGA)is an approved pharmaceutical excipient, which has been widely used in the field of medical materials due to its good biocompatibility and biodegradability.
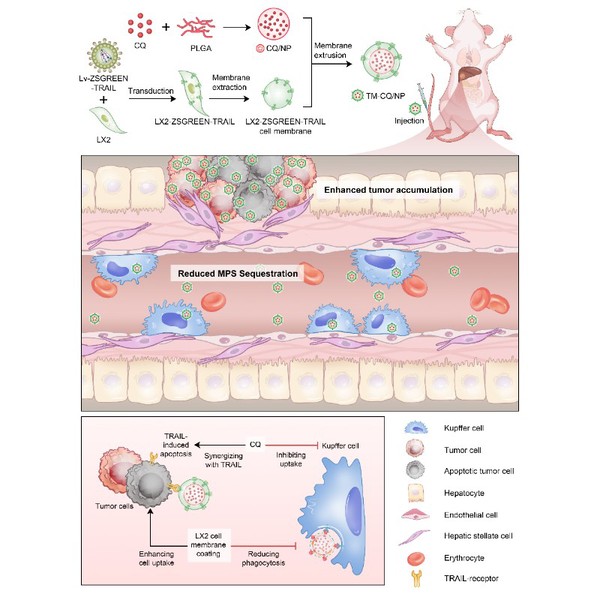
Figure 1. Schematic of the preparation of TM-CQ/NPs for macrophage-evading and effective cancer therapy.
Recently, Prof. LIU Xiangrui and Prof. ZHOU Tianhua from the Zhejiang University School of Medicine co-published an article entitled “Macrophage-evading and tumor-specific apoptosis inducing nanoparticles for targeted cancer therapy” in Acta Pharmaceutica Sinica B. In this study, the team developed a “cell & molecular” dual targeting biomimetic nanodrug that can effectively reduce the sequestration of mononuclear phagocyte system (MPS) and selectively induce apoptosis of tumor cells, using therapeutic proteins expressing fibroblasts membrane encapsulation strategy. PLGA nanoparticles (NP) were used as the model nanoparticles, and cell membrane coated nanoparticle (M-NP) was fabricated by membrane coating of activated hepatic fibroblasts. The endocytosis of M-NP by macrophages was reduced through the coated cell membrane presents CD47 to protect the nanoparticles from phagocytosis and the reduction of the formation of protein corona on M-NP. At the same time, the uptake of M-NP by tumor cells increased by the N/E-cadherin adhesion between tumor cell membrane and activated fibroblast cell membrane.
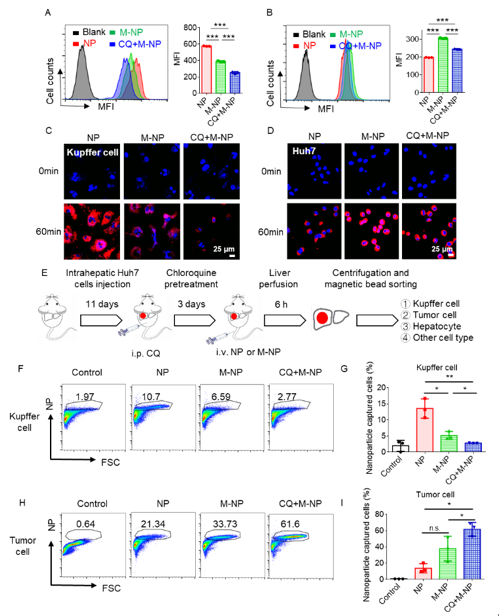
Figure 2. Chloroquine treatment improved the biodistribution of M-NPs and increased their internalization in tumor cells.
Considering the enhanced potential of nanoparticles to accumulate in the tumor tissue by membrane coating, the functionalization of cell membrane with the expression of therapeutic proteins could generate potent anticancer effect. The team constructed tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) stably expressing hepatic fibroblast cell line and TRAIL-expressing cell membrane (TM). TM-coated nanoparticles (TM-NP) can induce apoptosis of tumor cells through the specific binding of TRAIL death receptor, but have limited effect on normal cells. Chloroquine (CQ), a clinical autophagy inhibitor, was encapsulated in TM-NP to fabricate TM-CQ/NP. CQ could further inhibit the uptake of M-NP by macrophages, and synergize with TRAIL protein to induce tumor cell apoptosis.
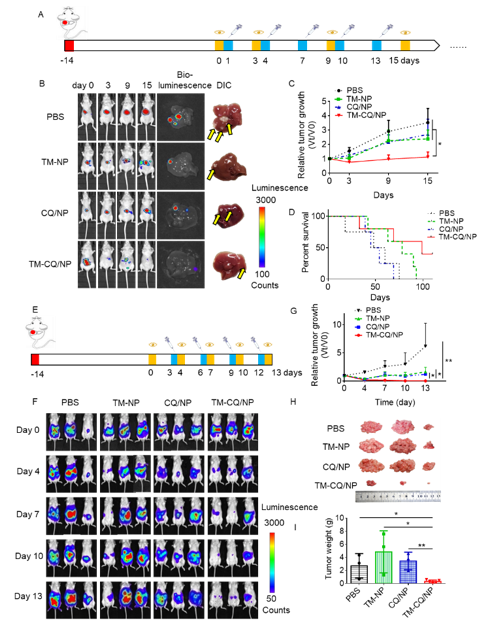
Figure 3. TM-CQ/NPs inhibited the tumor growth in orthotropic HCC tumor model and intraperitoneal tumor models.
This nanosystem presented dramatic antitumor activity in both orthotopic liver and intraperitoneal tumor models, indicating the therapeutic potential of our macrophage-evading and tumor-targeted cancer therapeutic strategy.
Source: School of Medicine, Zhejiang University