ZJU scientists identify O-GlcNAcylation as cause of pancreatic tumor growth by regulating MDH1
Pancreatic cancer is an exceedingly malignant tumor in the digestive system, and its five-year survival rate is little more than 10%. Metabolic alterations are one of the hallmarks of tumor cells. Oncogenic Kras-activated pancreatic ductal adenocarcinoma (PDAC) cells depend heavily on an unconventional glutamine (Gln) catabolic pathway to sustain cell growth. In the conventional pathway, Gln is first converted to aspartate (Asp), which is transported from mitochondria into the cytosol, where it is converted sequentially by aspartate transaminase 1 (GOT1), MDH1 and malic enzyme 1 (ME1) to pyruvate and NADPH. This pathway is critical for PDAC cells to maintain redox homeostasis and is required for cellular proliferation and tumor growth in vivo. Therefore, an insightful understanding of this regulatory mechanism may well open up a new avenue for the clinical treatment of PDAC.
The research team led by Prof. ZHOU Ruhong and Prof. YI Wen from the Zhejiang University College of Life Sciences published an article in the journal Nature Chemical Biology on July 25. This article reveals that O-GlcNAcylation contributes to pancreatic cancer growth by regulating the metabolic activity ofmalate dehydrogenase 1 (MDH1). This finding has immense implications for the development of drugs for pancreatic tumor.
The team have conducted relevant research. Their study demonstrates that Kras mutation induces cellular O-linked β-N-acetylglucosamine (O-GlcNAc), a prevalent form of protein glycosylation. Malate dehydrogenase 1 (MDH1), a key enzyme in the glutamine catabolic pathway, is positively regulated by O-GlcNAcylation on serine 189 (S189). Molecular dynamics simulations suggest that S189 glycosylation on monomeric MDH1 enhances the stability of the substrate-binding pocket and strengthens the substrate interactions by serving as a molecular glue. Depletion of O-GlcNAcylation reduces MDH1 activity, impairs glutamine metabolism, sensitizes PDAC cells to oxidative stress, decreases cell proliferation and inhibits tumor growth in nude mice. Furthermore, O-GlcNAcylation levels of MDH1 are elevated in clinical PDAC samples.
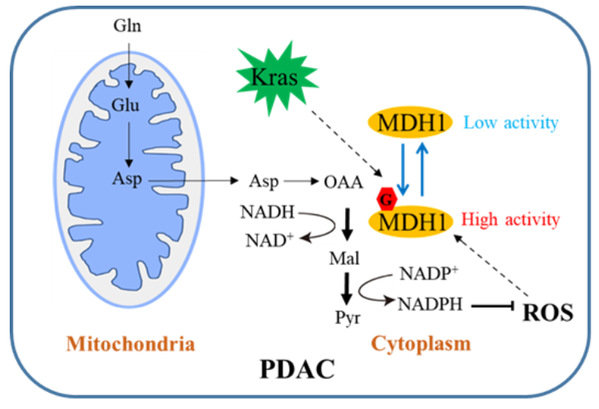
Schematic diagram of how MDH1 glycosylation regulates PDAC growth
The identification of specific pathways and proteins with unique dependency on O-GlcNAc is indispensable for developing targeted therapies. The study by Prof. Zhou and Prof. Yi et al. reveals that MDH1 glycosylation is poised to regulate the unique Gln metabolic pathway in PDAC, thus highlighting the potential to intervene in MDH1 glycosylation as a therapeutic strategy against PDAC.