How does bird flu jump to humans? A team from Zhejiang University finds a key clue
Bird flu, SARS, HIV/AIDS … Many of the infectious diseases that have unsettled the world have one thing in common: they first emerged in animals. Birds, bats, monkeys and other hosts can carry viruses that, under certain conditions, cross the species barrier and infect humans. What makes such diseases so alarming is not only their impact but also their unpredictability. They often appear suddenly, evolve rapidly, and spread before clear warning signs emerge.
That is why scientists around the world are trying to answer an important question: how do viruses jump from animals to humans, and how can we stop them?
A research team from Zhejiang University has now found an important part of the answer. On February 27, a team led by Professor YU Xiaofang, Director of the Cancer Institute at the Second Affiliated Hospital of the Zhejiang University School of Medicine, published a paper titled “STING–NF-κB signaling builds an influenza spillover barrier” in Science. The paper reveals a key mechanism the human body uses to block avian influenza, or bird flu, from adapting to humans.

Looking back at history, several influenza pandemics were linked to avian-origin influenza A viruses. Even today, bird flu remains a serious public health threat around the world. Yet not every bird flu virus that enters the human body may cause infection. One major reason lies in the body’s innate immune system — the body’s first line of defense against invading pathogens.
Professor YU has devoted much of his career to studying infectious diseases. In his view, viruses are highly adaptable enemies. “These pathogens are extraordinarily clever,” he noted. “They have evolved many survival strategies that we still do not fully understand.”
So when a virus enters the body, how does the immune system know it is there? And what helps stop the virus before it spreads?
To answer these questions, the team focused on two major immune pathways: NF-κB and cGAS–STING. These names may sound complicated, but their job is simple to understand: they help the body sense danger and launch a defense.
The team found that during influenza infection, the cGAS–STING pathway can activate NF-κB, which then helps suppress the virus. Together, they form an important barrier that makes it harder for bird flu to gain a foothold in human cells.
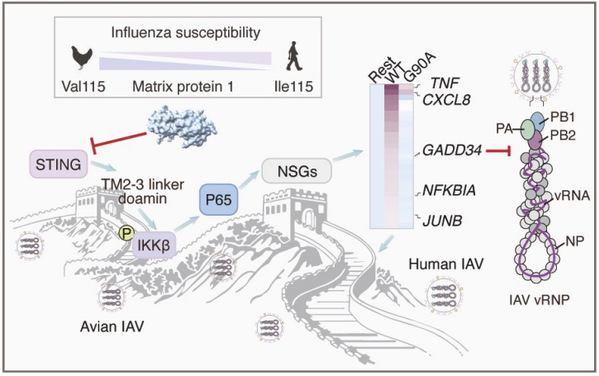
STING-NF-κB-NSGs: a host cell defense line against influenza
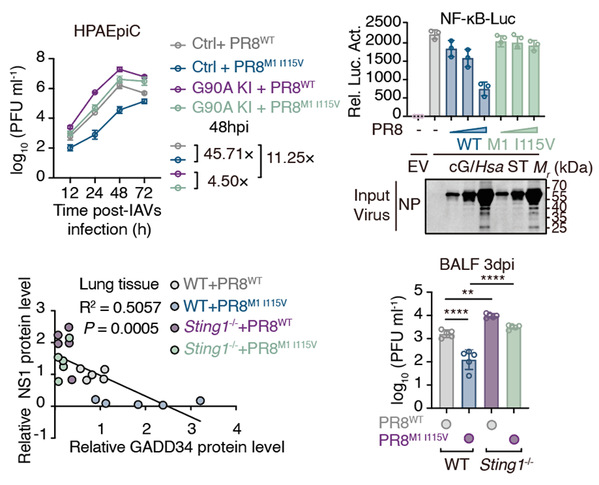
The team then identified a very small but very important detail in this process: a site called Gly90 in the STING protein. This tiny molecular position functions like a switch. When Gly90 is intact, the immune defense works well. But when this site is altered through the G90A mutation, the body becomes much less effective at holding back the virus. In other words, this tiny change weakens an important protective barrier. The researchers confirmed this result in both human airway cells and animal models.
The team went further and found a new group of antiviral genes controlled by NF-κB. They named them NF-κB–stimulated genes, or NSGs. Among them, one gene stood out: GADD34. The researchers found that GADD34 plays a particularly important role in suppressing influenza virus replication. Once the cGAS–STING pathway is activated, the Gly90 switch drives up GADD34 expression. GADD34 then interferes with the virus’s ability to reproduce inside cells.

Further experiments showed that GADD34 is highly active in the human respiratory tract, where influenza viruses usually attack first. It can block viral replication by targeting the virus’s polymerase activity — a key step the virus needs in order to multiply.
This finding is especially interesting because it shows that GADD34 has an antiviral function that scientists did not fully recognize before.
The study did not only look at how the human body fights viruses. It also examined how viruses fight back.If bird flu viruses are to infect humans successfully, they must learn how to get around the body’s immune defenses. To study this, the team analyzed influenza strains that are known to have crossed from animals to humans. They found that a viral protein called M1 plays an important role.
In human-adapted influenza viruses, the M1 protein is much better at blocking the STING–NF-κB–GADD34 defense pathway. In avian influenza viruses, it is much weaker. The team then identified a mutation that appears to enhance this adaptive ability: a change at amino acid 115 of the M1 protein, from valine to isoleucine. Although this change is small, it significantly improves the virus’s ability to resist the human immune response and replicate in human cells.
STING restricts the replication of avian site-carrying viruses
This discovery is not just important for basic science. It may also help public health experts identify dangerous bird flu strains earlier. If scientists can monitor key mutations in the M1 protein, they may be better able to judge which viruses circulating in birds have a greater chance of infecting humans. That could make surveillance more precise and improve early warning efforts.
The study reflects the close integration of clinical medicine and basic research at Zhejiang University. Drawing on the research and clinical platforms of the Zhejiang University Cancer Institute and the Second Affiliated Hospital, Professor YU’s team has built connections between oncology, immunology, and infectious disease prevention.
Their work offers a better understanding of how the body blocks avian influenza spillover. It also provides new ideas for monitoring pandemic risk and exploring treatments for other diseases, including tumors, autoimmune disorders and neurological conditions.

In the battle between viruses and humans, the spillover is often the moment when the balance shifts. By uncovering one of the body’s key defense mechanisms, the team has brought scientists a step closer to understanding and perhaps preventing the next outbreak.
Translator: FANG Fumin
Editor: HAN Xiao