A dual-functional RNA system reprograms antigen presentation to overcome tumor immune evasion
Immune checkpoint inhibitors (ICIs) are foundational in first-line treatment for hepatocellular carcinoma (HCC). However, low response rates remain a major clinical challenge. In advanced HCC, single-agent atezolizumab achieves an objective response rate (ORR) of only 17%, and combination therapy with bevacizumab reaches only 27.3–36%. Similarly, early-phase mRNA vaccine trials in advanced solid tumors also report a low ORR. A primary cause of immunotherapy resistance is tumor immune evasion, often achieved through the downregulation of major histocompatibility complex class I (MHC-I) molecules—which reduces cytotoxic T lymphocyte (CTL) recognition—and the loss of tumor antigens.
To address the dual mechanisms of MHC-I downregulation and antigen loss, on October 14, 2025, Prof. LING Qi from the The First Affiliated Hospital, Zhejiang University School of Medicine and Prof. LI Hongjun from Zhejiang University, School of Pharmacy co-published an article entitled “Reversal of tumour immune evasion via enhanced MHC-Class-I antigen presentation by a dual-functional RNA regulated system” in the journal Molecular Cancer. The research team designed a dual-functional RNA delivery system. The system utilizes lipid nanoparticles (LNPs), known for their liver-targeting capability, making them particularly suitable for HCC treatment. These LNPs co-encapsulate two key components (h-LNP): siRNA targeting proprotein convertase subtilisin/kexin type 9 (PCSK9) and mRNA encoding a model tumor antigen (ovalbumin, OVA). PCSK9 promotes lysosomal degradation of MHC-I molecules. Silencing PCSK9 is intended to stabilize MHC-I on the tumor cell surface.mRNA molecules supplement tumor antigens to counteract antigen loss.
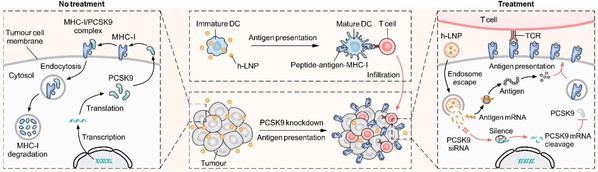
Figure 1 Schematic illustrating: PCSK9 mediated MHC-I degradation, PCSK9 knockdown elevating MHC-I level and epitope presentation, antigen encoding mRNA activating antigen specific T cells and promoting T cells infiltration into tumors.
Clinical and Mechanistic Validation: Analysis of patient samples solidified the link between low MHC-I expression and poor prognosis in HCC. Bioinformatics analysis further identified PCSK9 as the only MHC-I regulator significantly correlated to HCC patient survival, pinpointing it as a promising therapeutic target.
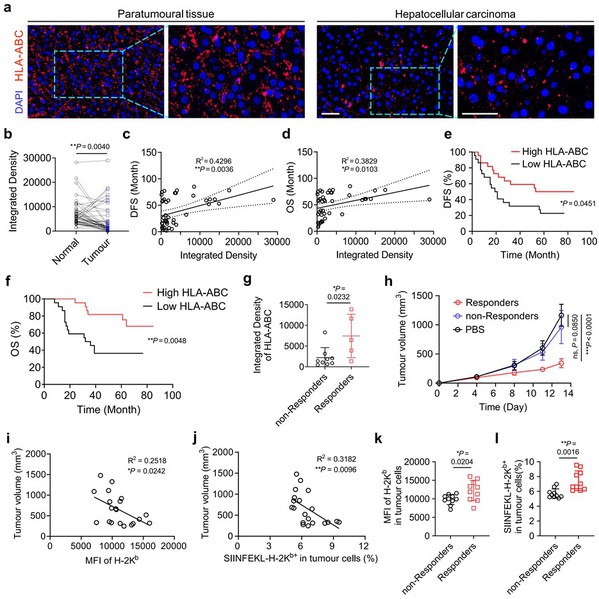
Figure 2. The correlation between MHC-I gene expression and prognosis as well as immunotherapy responsiveness.
Potent Anti-Tumor Activity of h-LNP In Vivo: In ectopic HCC tumors, h-LNP reduced final tumor volume to one-third of that observed in groups treated with siRNA (si-LNP) or mRNA (m-LNP) alone, with no adverse effects on body weight.
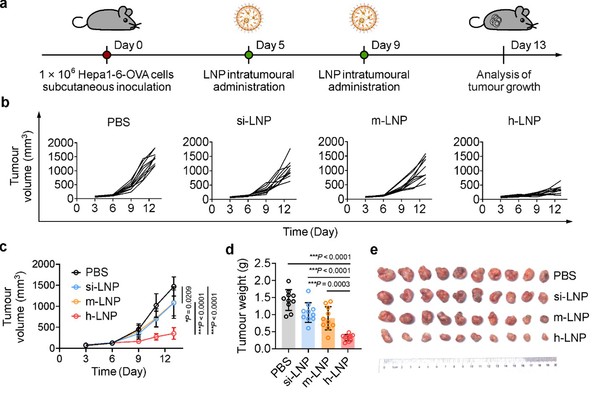
Figure 3. h-LNP treatment retards tumor growth.
Reconfiguration of the Tumor Microenvironment: scRNA-seq and immunophenotyping revealed that h-LNP treatment fundamentally remodeled the TME from an immunosuppressive to an immunologically active state. Tumors showed elevated infiltration of CD45+ immune cells, with significant increases in the proportions of CD8+ T cells and natural killer (NK) cells. The number of IFN-γ-secreting CD8+ T cells nearly doubled compared to the mRNA-only treatment group.
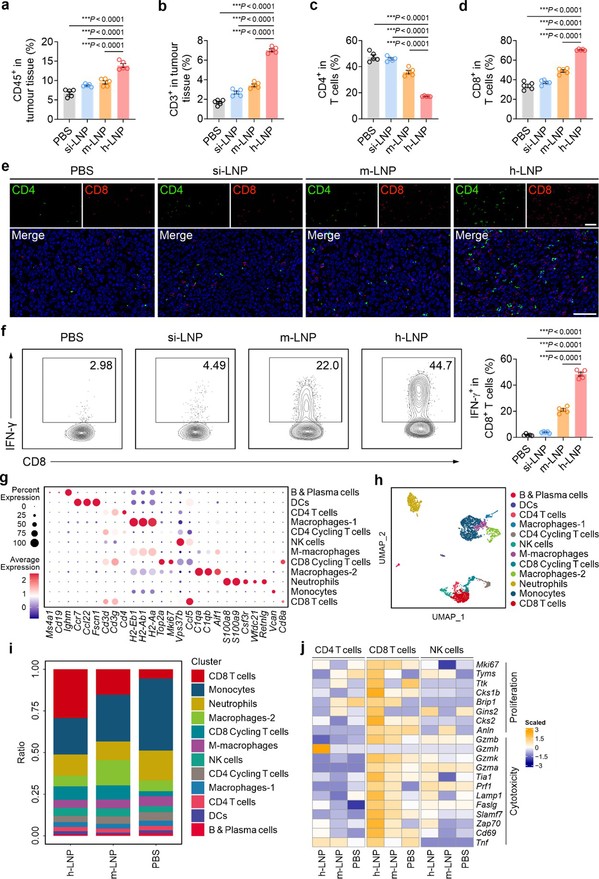
Figure 4. h-LNP treatment reprograms immune microenvironment in the tumor.
Overall, this study develops a LNP-delivered dual-functional RNA system that simultaneously inhibits MHC-I degradation and supplements tumor antigens. It overcomes one of key resistance mechanisms-antigen presentation disorder-in HCC immunotherapy, validates PCSK9 as a promising target, and provides a translatable strategy for optimizing mRNA vaccines. The findings hold significant potential for improving the response rates of mRNA vaccine in HCC immunotherapy.
More information: Dr. MENG Chaoyang, ZHANG Huipeng, YI Xuewen, and KONG Gangcheng are the co-first authors of this article. Prof. LING Qi and Prof. LI Hongjun are the co-corresponding authors of this article.
Source: The First Affiliated Hospital, Zhejiang University School of Medicine
Editor: HAN Xiao





