ZJU researchers discover a new mechanism for gastric cancer metastases
Recently, the research team headed by Prof. ZHOU Tianhua from the School of Medicine, Zhejiang University, published an article entitled “Long Non-coding RNA GMAN, Upregulated in Gastric Cancer Tissues, is Associated with Metastases in Patients and Promotes Translation of Ephrin A1 by Competitively Binding GMAN-AS” in the journal of Gastroenterology. They identified long non-coding RNAs (lncRNAs) that are upregulated in gastric cancer tissues from patients and did research into their function in gastric tumor metastases.
Gastric cancer is one of the most prevalent malignant tumors all over the world and ranks among the leading causes of mortality. In China, the incidence and mortality rate of gastric cancer comes in second among all malignant tumors. Research reveals that the 5-year survival rate of patients diagnosed with gastric cancer is approximately 25% and that 40% of the patients suffer from gastric cancer metastases. However, the biological process and the molecular mechanism regarding gastric cancer metastases remain obscure. It is therefore of considerable significance to delve into the molecular mechanism for gastric cancer metastases and seek for effective therapy for gastric cancer.
In microarray analyses, ZHOU Tianhua et al. identified 151 lncRNAs expressed at significantly higher levels in gastric tumor vs. non-tumor tissues. Levels of an lncRNA termed as gastric cancer metastases associated long non-coding RNA (GMAN) increased in gastric tumor tissues, compared with non-tumor tissues; its upregulation was related to tumor metastases and a shorter survival time of patients. The GMAN gene overlaps with the ephrin A1 gene (EFNA1) and was highly expressed in BGC-823 and MKN45 cells. Knockdown of the GMAN in these cells did not affect proliferation, colony formation, or adhesion, but did reduce their invasive activity in transwell assays. Ectopic expression of GMAN increased the invasive activity of HGC-27 cells. BGC-823 and MKN45 cells with knockdown of GMAN formed fewer metastases following injection into tail veins of nude mice. Knockdown or knockout of GMAN also reduced levels of EFNA1 protein in cells. They found that GMAN promoted translation of EFNA1 mRNA into protein by binding to the antisense GMAN RNA (GMAN-AS)—this antisense sequence is also complementary to that of EFNA1 mRNA. Levels of EFNA1 protein also increased in gastric tumors from patients with metastases, compared to that without metastases. Knockout of EFNA1 in BGC-823 cells reduced their invasive activity in transwell assays and ability to form metastases following injection into SCID mice. Ectopic expression of EFNA1 in BGC-823 cells with knockdown or knockout of GMAN restored their invasive activities and ability to form metastases in nude or SCID mice. A CRISPR/Cas9-based strategy to disrupt the GMAN gene significantly reduced the numbers of metastases formed from SGC-7901 cells in mice.
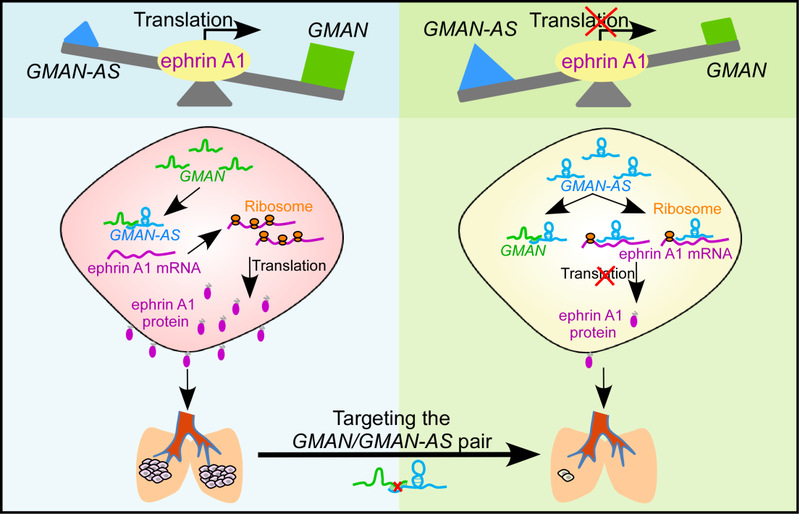
To recap briefly, this research identifiedan lncRNA which can regulate the translation of EFNA1 mRNA by binding competitively to GMAN-AS RNA, revealed a novel mechanism for the regulation of gastric cancer metastases and explored the prospects for the prevention or reduction of gastric cancer metastases by adopting a CRISPR/Cas9-based strategy.





