Can heart failure treatment break new ground? Zhejiang University scientists publish breakthrough study in Nature
The heart is often called the body’s “tireless pump,” working around the clock to keep us alive. But like any machine, it will wear down. When the heart becomes too weak to pump blood effectively or too stiff to fill properly, the body can no longer get the oxygen and nutrients it needs. This condition is known as heart failure.
Heart failure is a major global health challenge. According to the World Health Organization, more than 64 million people around the world are living with it. Even with today’s standard treatments, severe heart failure still carries a five-year mortality rate of nearly 50 percent. This harsh reality reflects an urgent need for better therapies that can target the disease more effectively.

A new study from Zhejiang University may offer fresh hope. Published in Nature on April 8, the research reports a promising new approach to treating heart failure. Titled “Engineered immunosuppressive dendritic cells protect against cardiac remodelling,” the study was led jointly by Professor HU Xinyang’s team and Professor XU Yang’s team. Their work represents an important step forward in the search for more effective treatments for heart failure.
Recalling what first inspired the research, HU Xinyang spoke about a young patient she met several years ago. The man, in his twenties, was suffering from dilated cardiomyopathy and had already developed severe heart failure by the time he came to the clinic. His condition kept deteriorating, standard medications were no longer working well, and the possibility of a heart transplant felt painfully remote.
“What I remember most is his father, who came with him and asked the same desperate questions again and again, crying in my office,” HU Xinyang said. “At that moment, I kept asking myself: apart from the existing options, could there be any alternative way to treat terminal heart failure?”

As a cardiologist, HU Xinyang knew that ongoing immune-driven inflammation plays a major role in making heart failure worse. After a heart attack or in chronic heart failure, the body’s immune response can become overactive. Instead of helping with repair, it may continue attacking the heart muscle, driving scarring and structural damage. HU Xinyang compares this process to “guards who have lost control,” turning their weapons on the very organ they are meant to protect.
This is also why treating inflammation in heart failure has been exceedingly difficult. Existing anti-inflammatory therapies often bring only limited or short-lived benefits. In some cases, they may even suppress the body’s normal protective immune responses and raise the risk of infection.
Years of clinical practice, together with her sensitivity to patients’ unmet needs, made HU Xinyang come to the realization that if scientists could find a way to better control this harmful immune response while protecting the heart function, it might open the door to a new treatment path for many patients.
HU Xinyang reached out to her colleague, immunologist XU Yang, who has devoted over 30 years to studying how the immune system works. Over the years, XU Yang has made a number of internationally recognized contributions to the field of immune regulation. When he heard about the clinical challenges that doctors were faced with, he immediately saw both the urgency and the potential. The two quickly found common ground and decided to tackle the problem together.

They brought together a team full of curiosity and drive. “We met every three days, no matter what,” XU Yang recalled. “And at key moments, we discussed ideas and results every single day.” It was this constant exchange of ideas that helped move the research forward.
For patients with end-stage heart failure, one of the biggest challenges is that doctors still cannot precisely control the underlying inflammation. When inflammation in the heart remains unchecked, it gradually leads to fibrosis that cannot be reversed. Over time, the heart weakens further and further. Many patients go through repeated hospital stays, with few effective options left. This problem has long frustrated cardiologists around the world.
The team focused on dendritic cells, often described as the “commanders” of the immune system because they help direct immune responses. After nearly five years of hard work, they developed a new approach: engineered dendritic cells that can both suppress harmful immune activity and find their way directly to damaged heart tissue. They call this strategy iCDC.
“In a way, this is a true ‘from zero to one’ breakthrough,” the team said. “It opens up a completely new direction for treating heart failure.”

Using gene-editing technology, the researchers essentially gave these cells a built-in “GPS” so they can home in on the heart. At the same time, they programmed the cells to maintain a calming, anti-inflammatory state. Put simply, these immune “commanders” are sent into the heart with clear instructions: quiet the harmful, ongoing inflammation and create the right conditions for healing.
XU Yang noted that research on dendritic cells (DCs) was recognized with the Nobel Prize back in 2011. “But despite years of work around the world, it hasn’t been easy to find the right way to use them in real treatments,” he said. While DCs are often used to activate the immune system, such as in cancer therapies, the team took a different path here, using them instead to dial down an overactive immune response in heart failure.
HU Xinyang explained,“What makes iCDC especially promising is that it can regulate the immune response in the damaged heart without suppressing the immune system throughout the whole body. That means a much lower risk of side effects like infections, and a higher level of safety.”
In a series of animal studies, the results were encouraging. In mouse models of conditions such as heart attack and long-term cardiac stress, the therapy helped reduce harmful fibrosis in the heart, improve blood flow, strengthen the heart’s pumping ability, and ultimately increase survival. Even more promising, similar benefits and safety were also confirmed in studies with non-human primates.
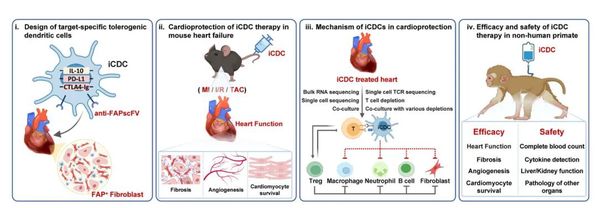
Schematic of iCDC engineering and the experimental workflow. Therapeutic efficacy and mechanisms were evaluated in mouse models of MI, I/R injury and TAC, and translational efficacy and safety were assessed in an NHP MI model.
This breakthrough didn’t happen overnight. It is the outcome of years of persistence, as well as a strong and supportive research environment. At Zhejiang University, close collaboration across disciplines, together with rich clinical resources from its affiliated hospitals, has created what researchers describe as a “fast track” from lab discoveries to real-world medical applications.
“We’ve always believed that real clinical problems should guide our research and that our findings should benefit clinical practice,” HU Xinyang said. In this project, her team placed particular emphasis on safety and real-world applicability, pushing the research forward with both creativity and care.
XU Yang shared a similar vision. “I’ve always hoped that our work could go beyond the lab and truly make a difference for patients,” he said. “Here, we have the rare advantage of a complete research system from basic science to advanced animal studies, which allows us to take important steps toward clinical use.”

Looking ahead, the two teams will continue working closely to refine how these engineered cells are produced, with the goal of bringing the therapy into clinical trials as soon as possible.
Behind this research lies not only scientific expertise, but also deep compassion, curiosity, and determination. Starting from real challenges faced in the clinic, the team has opened up a new possibility for treatment. And their goal is crystal clear: to turn this promising approach into a safe, effective therapy that can one day bring new hope to millions of people living with heart failure.
Translator: FANG Fumin
Editor: HAN Xiao





