Researchers discover T cells use mechanical force to “hook” cancer cells
Immunotherapy, as one of the most promising cancer treatments, boosts the body’s immune system to fight cancer by activating T cells. T-cell receptors (TCRs) on T cell surface specifically and sensitively recognize rare foreign antigen or tumor neoantigen presented by major histocompatibility complexes (pMHCs) on cancer cells, which plays a vital role in recognizing and killing cancer cells. However, it still remains enigmatic that how to improve and control the efficiency of T cell killing in immunotherapy. The most important and difficult question is to uncover the detailed mechanism of TCR antigen recognition.
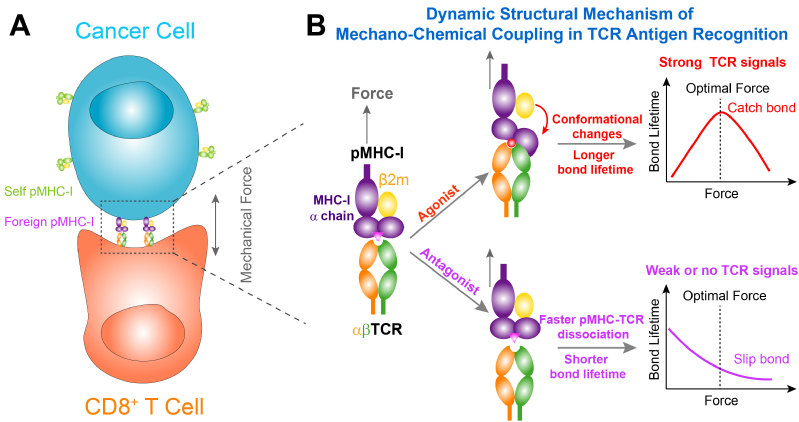
Recently, the research team led by Prof. CHEN Wei in ZJU's School of Medicine, in collaboration with the research team led by Dr. LOU Jizhong in the CAS Institute of Biophysics, published a research article entitled “Mechano-regulation of peptide-MHC class I conformations determines TCR antigen recognition” on Molecular Cell. They report that a dynamic structural mechanism of mechano-chemical coupling for TCR antigen recognition. They found mechanical force-induced conformational changes in the agonist peptide-MHC-I—allosterically activates TCR-pMHC-I catch bonds to determine TCR antigen recognition and trigger T cell signaling. The lifetimes of TCR-pMHC bond under force are measured at the single-molecule level through biophysical experiments.

The accurate and fast recognition and removal of infected cells or genetically mutated cancer cells is essential to the maintenance of a healthy body. CD8+ tumor-infiltrating T cells (TILs) play an indispensable role in this process.TCRs on CD8+ T cells can specifically recognize tumor neoantigen to initiate T cell signaling and adaptive immunity rapidly. Nonetheless, there are a massive of “non-self” antigens in human bodies, which have very little differences with “self” antigens with merely several amino acid residues.
How TCRs can discriminate “non-self” antigens in an ocean of “self” antigens in a rapid and precise manner? This is not only one of the important questions in the field of immunology but also one of the keys to T cell-based immunotherapy, particularly TCR-T.
The T cell has no eyes. Then how can it recognize “self” and “non-self” cells? In an article published by Prof. CHEN Wei in the journal of Cell in 2014, they found that force-induced TCR-pMHC catch bonds (force prolongs bond lifetime) amplify the discriminative power of TCRs for “self” and “non-self” antigens.
Video of SMD Simulation Trajectory for Pulling the dEV8-H-2Kb-2C-TCR Complex
In this study, Prof. CHEN Wei et al.further investigate the molecular mechanism of the TCR catch bond. They integrate steered molecular dynamics (SMD) simulation, single-molecule biophysical approaches, and functional assays to collectively demonstrate that mechanical force induces conformational changes in pMHCs to enhance pre-existing contacts and activates new interactions at the TCR-pMHC binding interface to resist bond dissociation under force, resulting in TCR-pMHC catch bonds and T cell activation. Under force, mechanical pulling on TCR-pMHC-I complex can induce rotational conformational changes of α1-α2 domains in MHC-I by disrupting the intramolecular interactions between β2 microglobulin and α3 or α1-α2 domains. This rotation enhances TCR-pMHC-I binding by forming new hydrogen bonds within the interface between TCR and pMHC-I. Intriguingly, cancer-associated somatic mutations in HLA-A2 that may restrict these conformational changes suppress TCR-pMHC catch bonds. Structural analysis also indicates that HLA polymorphism might alter the equilibrium of these conformational changes. These findings not only reveal critical roles of force-induced conformational changes in pMHCs for activating TCR-pMHC catch bonds but also have implications for T cell-based immunotherapy.





