Ubiquitin ligase promotes pro-inflammatory mononuclear phagocytes, exacerbating intestinal inflammation
The status quo in a healthy gut is related to a constant balance between the resident macrophages and inflammatory mononuclear phagocytes (Mphs) that is critical for maintaining and ensuring protective immunity. How colonic microenvironment influences the inflammatory MPh infiltration is not well defined and represents a fundamental gap in the understanding of homeostatic immune function and Inflammatory Bowel Disease (IBD) development
The research team led by Dr. WANG Qingqing with the Zhejiang University Institute of Immunology published an article entitled “Fbxw7 increases CCL2/7 in CX3CR1hi macrophages to promote intestinal inflammation” online in the July 27 issue of the Journal of Clinical Investigation. This study reveals a novel function of E3 ligase Fbxw7 in regulating intestinal inflammation.
Resident and inflammatory mononuclear phagocytes (MPh) in the intestine are critically involved in the pathology of Inflammatory Bowel Diseases (IBD). CX3CR1hi (CX3CR1 high-positive) resident macrophages can contribute to the maintenance of gut homeostasis and protect the host from certain pathogens. In inflammation environments, the concentration of numerous Ly6Chi (Ly6C high-positive) monocytes in specific intestinal sections will give rise to CX3CR1int (CX3CR1 intermediate-positive) MPh, thereby aggravating inflammation by activating Th17 and innate lymphoid cells. Research into the precise regulation of the tendency, accumulation and division of phagocytes in damaged intestines may provide insight into the mechanism and treatment of IBD.
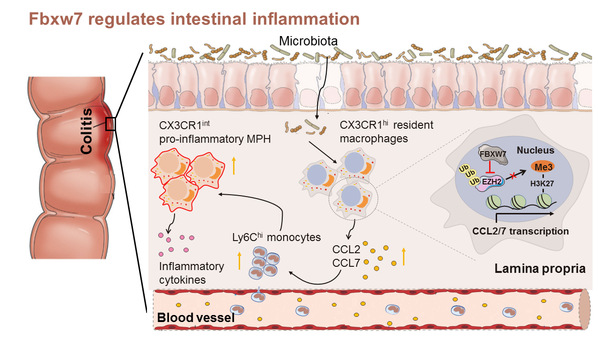
E3 ligase Fbxw7 is a vital cancer-inhibiting gene, but its function in IBD remains elusive. WANG Qingqing et al. discover that increased expression of E3 ligase FBXW7 in the inflamed intestine is significantly correlated to IBD severity in both human diseases and mice models. Myeloid-Fbxw7 deficiency protectes mice from dextran sodium sulfate (DSS) and 2,6,4-trinitrobenzene sulfonic acid (TNBS) induced colitis. Fbxw7 deficiency results in decreased production of chemokines CCL2 and CCL7 by colonic CX3CR1hi resident macrophages and reduced accumulation of CX3CR1int pro-inflammatory MPh in colitis colon tissues. Mice which receive AAV-shFbxw7 administration exhibita significantly improved survival rate and alleviated colitis. Mechanisms screening demonstrates that FBXW7 suppresses H3K27me3 modification and promotes Ccl2 and Ccl7 expression via degradation of histone-lysine N-methyltransferase EZH2 in macrophages.

This study indicates that FBXW7 degrades EZH2 and increases Ccl2/Ccl7 in CX3CR1hi macrophages, which promotes the recruiting CX3CR1int pro-inflammatory MPh into local colon tissues with colitis. Targeting FBXW7 may offer a potential therapeutic approach to intestinal inflammation intervention.





