Gold-silver nanoshells promote wound healing from drug-resistant bacteria infection, ZJU researchers find
Chronic infections have gradually become prevalent in clinical practice. If underappreciated, they may develop into systemic infections, septicemia and multiple organ dysfunctions. The presence of multidrug-resistant bacteria in operating rooms or complex outdoor environments will further exacerbate this challenge. Traditional antibiotic therapy fails to cater to the rapidly increasing clinical needs in terms of effectiveness and costs. Therefore, there is a compelling need to develop novel antibacterial approaches and materials. In addition, if residual bacteria on the wound can be precisely monitored in real time, it will provide a more effective way for doctors to administer treatment at the optimal time.
The research team led by ZHOU Min from the Second Affiliated Hospital and the Institute of Translational Medicine of Zhejiang University explored a prospective approach to overcome these challenges through gold-silver nanoshells (AuAgNSs). They adopted Surface-Enhanced Raman Scattering Imaging to pinpoint infectious wounds and promote healing. Their findings were published in a research article entitled “Gold-Silver Nanoshells Promote Wound Healing from Drug-Resistant Bacteria Infection and Enable Monitoring via Surface-Enhanced Raman Scattering Imaging” in the journal of Biomaterials on January 18.
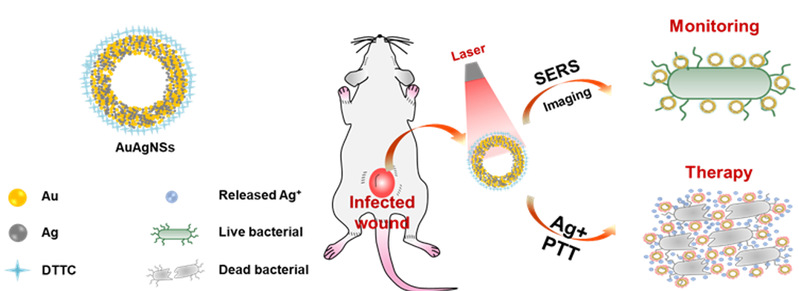
Researchers discovered that with marked responsiveness of the near-infrared laser due to surface plasmon resonance (SPR) from the hybrid metallic nanoshell structure, AuAgNSs exhibit a remarkable photothermal effect and they release silver ions during laser irradiation to bacterial eradication simultaneously. Two MDR bacteria strains, methicillin-resistant Staphylococcus aureus (MRSA) and extended-spectrum β-lactamase Escherichia coli, were chosen as models and studied both in vitro and in vivo. As a result, the AuAgNSs-DTTC substrates enabled surface-enhanced Raman scattering imaging to provide a non-invasive and extremely high sensitive detection (down to 300 CFU mL−1 for MRSA) and prolonged tracking (at least 8 days) of residual bacteria. In a chronic MRSA-infected wound mouse model, the AuAgNSs gel-mediated photothermal therapy/silver-release led to synergistic would healing with negligible toxicity or collateral damage to vital organs.
This study suggests that AuAgNSs-DTTC is a promising anti-bacterial tool for clinical translation.





