Joint effort of ZJU and CityU researchers show the role of Wdr26 in regulating nuclear condensation and enucleation
Red blood cells, as the most abundant type of cells, are primarily responsible for oxygen transportation in the human body. Mammalian red blood cells lack nuclei. Although nuclei can still be found in the red blood cells of such vertebrates as birds and fish, they appear to be very small at the maturation stage. In recent years, studies have showed that nucleoprotein degradation and nuclear concentration are the essential prerequisites for erythroblasts, but the mechanism of regulating erythrocyte nucleoprotein homeostasis remains elusive.
The research team led by CHEN Caiyong from Zhejiang University and the research team led by SHI Jiahai from the City University of Hong Kong conducted collaborative research into the role of Wdr26 in regulating nuclear condensation. Their findings were published in a cover article entitled “Wdr26 regulates nuclear condensation in developing erythroblasts” in the journal of Blood on January 17.
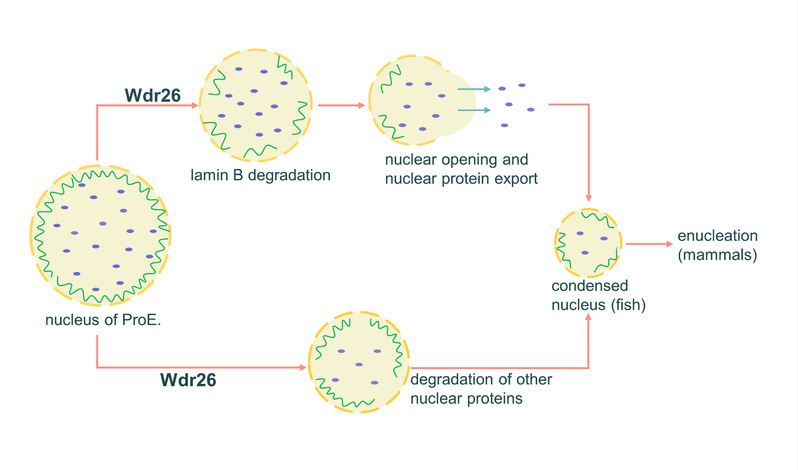 In their study, researchers found that Wdr26, a gene upregulated during terminal erythropoiesis, played an essential role in regulating nuclear condensation in differentiating erythroblasts. Losses of Wdr26 induced anemia in zebrafish and enucleation defects in mouse erythroblasts because of impaired erythroblast nuclear condensation. As part of the glucose-induced degradation-deficient ubiquitin ligase complex, Wdr26 regulated the ubiquitination and degradation of nuclear proteins, including lamin B. Failures of lamin B degradation blocked nuclear opening formation, leading to impaired clearance of nuclear proteins and delayed nuclear condensation.
In their study, researchers found that Wdr26, a gene upregulated during terminal erythropoiesis, played an essential role in regulating nuclear condensation in differentiating erythroblasts. Losses of Wdr26 induced anemia in zebrafish and enucleation defects in mouse erythroblasts because of impaired erythroblast nuclear condensation. As part of the glucose-induced degradation-deficient ubiquitin ligase complex, Wdr26 regulated the ubiquitination and degradation of nuclear proteins, including lamin B. Failures of lamin B degradation blocked nuclear opening formation, leading to impaired clearance of nuclear proteins and delayed nuclear condensation.
This study reveals an unprecedented role of an E3 ubiquitin ligase in regulating nuclear condensation and enucleation during terminal erythropoiesis. The results provide mechanistic insights into nuclear protein homeostasis and vertebrate red blood cell development.





