Nicotinamide shown to reverse sorafenib-induced HFSR
Sorafenib is a multikinase inhibitor that targets the receptor tyrosine and serine/threnonine kinases involved in tumor progression and tumor angiogenesis. It has been approved in many countries as a frontline drug for patients with advanced hepatocellular carcinoma, advanced renal cell carcinoma, and metastatic thyroid carcinoma. For most of these patients, sorafenib is the sole treatment able to produce favorable therapeutic effects. However, its association with a high incidence of hand–foot skin reaction (HFSR) hampers the clinical application of this anticancer drug. Clinical trial reports from ClinicalTrial.gov and other published papers have demonstrated that the incidence percentage of sorafenib-induced HFSR ranged from 30% to 76%. For patients with severe HFSR, dose reduction or interruption is needed, which undesirably reduces the therapeutic efficiency of sorafenib or even unlooses the progression of cancer.
Sorafenib-induced HFSR is characterized by hyper-keratosis in palmar and plantar areas. Hyper-keratosis, defined as stratum corneum thickening, generally results from the abnormality in epidermal homeostasis of keratinocytes, including hyper-proliferation and hyper-differentiation. Since numerous studies have uncovered that most drug-induced cutaneous toxicities are the after effects of pathways directly affecting keratinocytes, keratinocyte dysfunction has thus been largely speculated as the primary cause of sorafenib-induced HFSR.
However, intervention strategies based on this concept prove ineffective. For instance, controlling the presence of plantar hyper-keratosis by prophylactic removal of the hyper-keratotic areas followed by the application of a moisturizing cream has little effect on severe HFSR (grade II or III). Moreover, patients with HFSR could hardly benefit from the cushioning of callused areas by means of soft or padded shoes. Therefore, dose reduction or interruption is essential for patients developing high-grade HFSR, which inevitably circumscribes the therapeutic outcomes of sorafenib. The development of effective interventions based on the underlying mechanism of sorafenib-induced HFSR becomes urgently desirable.
Against this backdrop, Prof. YANG Bo et al. engage in relevant research. Their research findings are published online in the April 15 issue of Cell Research.
Their study demonstrates that vascular endothelial cells are the primary cellular target of sorafenib-induced HFSR wherein soluble heparin-binding epidermal growth factor (s-HBEGF) mediates the crosstalk between vascular endothelial cells and keratinocytes.
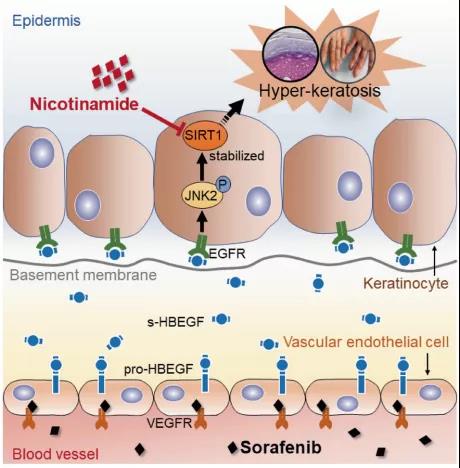
Mechanistically, s-HBEGF released from vascular endothelial cells activates the epidermal growth factor receptor (EGFR) on keratinocytes and promotes the phosphorylation of c-Jun N-terminal kinase 2 (JNK2), which stabilizes sirtuin 1 (SIRT1), an essential keratinization inducer, and ultimately gives rise to HFSR. The administration of s-HBEGF in vivo could sufficiently induce hyper-keratinization without sorafenib treatment.
Furthermore, researchers also find that HBEGF neutralization antibody, Sirt1 knockdown, and a classic SIRT1 inhibitor nicotinamide could all significantly reduce the sorafenib-induced HFSR in the mouse model. It is noteworthy that nicotinic acid, a prodrug of nicotinamide, could substantially reverse the sorafenib-induced HFSR in ten patients in a preliminary clinical study.
This study reveals the mechanism of vascular endothelial cell-promoted keratinization in keratinocytes and provides a potentially promising therapeutic strategy for the treatment of sorafenib-induced HFSR.





