A molecular clue to lifelong health: scientists uncover how fetuses keep growing under low-sugar conditions
What if one of the earliest secrets to lifelong health is hidden in the liver before birth?
Scientists from Zhejiang University and Xiamen University have uncovered a biological mechanism that helps fetuses keep growing even in a naturally low-sugar environment in the womb. Their findings shed light on how the body protects development in early life and how those same protective mechanisms may also shape health decades later.
The study, published on March 13, 2026, in Vita, was led by Prof. HUANG Hefeng from the School of Medicine at Zhejiang University and Prof. LIN Shengcai from the School of Life Sciences at Xiamen University. It was also the first original research article to appear on the journal’s newly launched website.
Before birth, a baby depends entirely on the placenta for nutrients from the mother. But the placenta also consumes some of the glucose itself, meaning that a fetus naturally lives in a relatively low-blood-sugar environment.
For adult liver cells, low glucose is usually a warning sign. When sugar levels fall, a cellular energy sensor called AMPK sounds the alarm, and another key growth regulator, mTORC1, is switched off to conserve energy.
In the fetal liver, however, the researchers found something strikingly different. Even when the low-glucose alarm is triggered, mTORC1 remains active, guiding protein production to continue at full speed. In other words, the fetal liver can keep building and growing, even under conditions that would normally force adult cells into the energy-saving mode.
This special metabolic protection appears to be rare and ephemeral. The researchers found that it does not occur in commonly used lab-grown cells such as HEK293T and MEF cells, or in liver cells from adult mice. In fact, the ability seems to be unique to fetal life and disappears rapidly after birth. Within about 24 hours, the liver shifts back to the standard adult pattern of responding to low glucose.
So how does the fetal liver do it? The answer lies in a signaling channel known as TRPV4.
In adult liver cells, TRPV4 is highly sensitive to low glucose levels. When glucose drops, the channel closes, setting off a cascade of signals that suppress growth-related activity. But in the fetal liver, TRPV4 behaves very differently: it stays open.
The researchers found that this is because an enzyme called p300 adds a chemical modification, known as acetylation, to the K608 site of the TRPV4 protein. That small molecular tag effectively locks the channel in an open state, preventing low-glucose signals from shutting down the growth machinery. As a result, mTORC1 can continue driving protein synthesis, helping the fetus maintain rapid development even when glucose is limited.
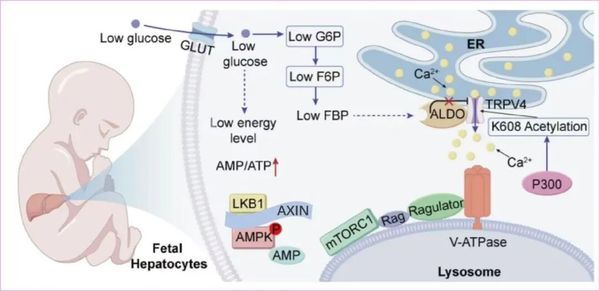
To test just how important this mechanism is, the team created transgenic fetal mice with a TRPV4-K608R mutation, which effectively disables this protective switch. The consequences were shocking: liver development was dramatically impaired, and some fetuses did not survive. The results suggest that acetylation at the K608 site of TRPV4 is essential for protecting fetal growth under low-glucose conditions.
Beyond its significance for developmental biology, the study may also have implications for long-term health. The researchers noted that while this early-life adaptation helps the fetus survive and grow in a resource-limited environment, it may also leave lasting metabolic “programming” in the body. From a life-course health perspective, molecular settings established before birth may serve as both an immediate survival strategy and a foundation for future health outcomes.
That raises an important possibility: if the prenatal environment is disrupted, for example, by maternal metabolic disorders that alter glucose levels, this fine-tuned protective system may become mismatched to its surroundings. Such early molecular imprints could persist for decades, potentially increasing the risk of obesity, diabetes, and other metabolic diseases later in life.
The discovery offers a new window into how the body safeguards growth at the very start of life and how those earliest adaptations may continue to influence health long after birth.
Translator: FANG Fumin
Editor: HAN Xiao





