ZJU scholars provide a novel AKI kidney-targeted agent
Acute kidney injury (AKI) manifests as a rapid decline in renal function with increased morbidity and mortality. The primary causes of AKI include kidney ischemia-reperfusion (IR) injury, sepsis, and nephrotoxins. The incidence of AKI has been on the rise, inducing the increased risks of chronic kidney disease and end-stage renal disease. The current treatment for AKI is still reliant on interventions such as early fluid resuscitation or renal replacement therapy, and there is a paucity of with effective pharmaceutical treatments. Renal mitochondria-targeted antioxidative therapy for AKI remains a challenge.
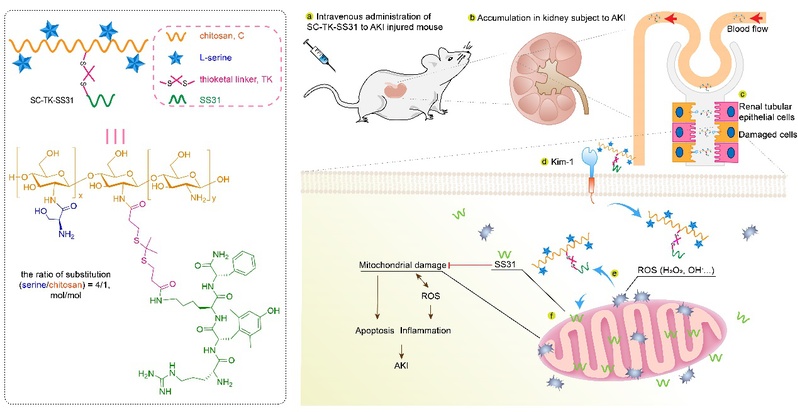
After about 2 years’ painstaking research, the research team led by Prof. DU Yongzhong from the Zhejiang University College of Pharmaceutical Sciences developed an AKI kidney-targeted drug which can be precisely delivered, effectively released, intensively accumulated and permanently sustained, thereby laying a solid foundation for clinical translation.Their research findings have appeared in the journal of Science Advances and captured the attention of Nature Reviews Nephrology.
The death of tubular epithelial cells (TEC) is the leading cause of AKI. Studies have shown that when AKI occurs, there is an abnormal increase in reactive oxygen species (ROS) in renal tubular epithelial cells, which acts as a trigger for the pathways toward cellular apoptosis, necrosis, and renal fibrosis.
This AKI process cannot be inhibited by drugs because conventional antioxidative therapy is unable to reach TECs precisely, and drugs will be excreted very quickly, for the kidney is a vital excretory organ of the body. Therefore, this therapy cannot cover and protect damaged mitochondria with a high concentration, thereby failing to stop the chain of destruction.
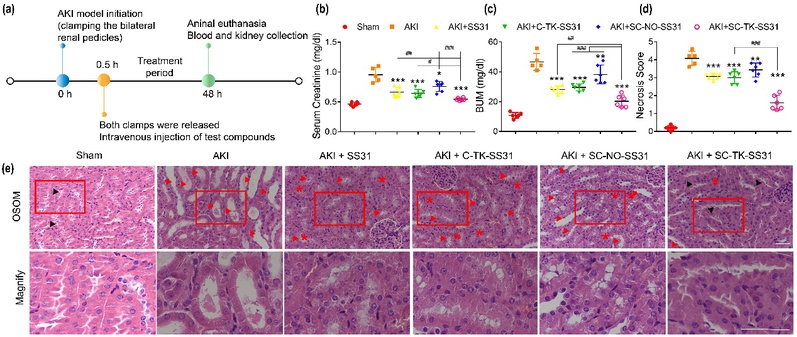
Against this background, Prof. DU Yongzhong et al. engaged in prodrug research in an attempt to provide a novel therapy for AKI. They synthesized L-serine–modified chitosan (SC) as a potential AKI kidney–targeting agent due to the native cationic property of chitosan and specific interaction between kidney injury molecule–1 (Kim-1) and serine. Results indicated that SC was rapidly accumulated and long-term retained in ischemia-reperfusion–induced AKI kidneys, especially in renal tubules probably due to the specific interactions between SC and Kim-1. SC-TK-SS31 was then prepared by conjugating SS31, a mitochondria-targeted antioxidant, to SC via reactive oxygen species (ROS)–sensitive thioketal linker. Because of the effective renal distribution combined with ROS-responsive drug release behavior, the administration of SC-TK-SS31 led to an enhanced therapeutic effect of SS31 by protecting mitochondria from damage and reducing the oxidative stress, inflammation, and cell apoptosis.
SC-TK-SS31 is therefore a promising candidate for the treatment of clinical AKI. The precisely stepwise targeting delivery strategy can also be applied to the treatment of other kidney diseases, such as renal cancer.





