Scientists discover a novel satiety sensor in the fly brain
The research team led by WANG Liming from the Zhejiang University Life Sciences Institute published a research article entitled “A novel satiety sensor detects circulating glucose and suppresses food consumption via insulin-producing cells in Drosophila” in the journal of Cell Research.
Sensing hunger and satiety is crucial to ensure appropriate and balanced intake of energy and essential nutrients for every animal species. Research reveals that several groups of hypothalamic neurons survey the organismal satiety state via a number of pathways including nutrients in the circulation system, gastrointestinal fullness and fat depots, and suppress food consumption accordingly. Impaired satiety sensing may contribute to the global epidemic of obesity and other related metabolic diseases in the human society. In mammalian models, several groups of hypothalamic neurons have been identified as satiety sensors, including those expressing pro-opiomelanocortin and those expressing melanocortin-4 receptor.
Fruit flies Drosophila melanogaster offer a simple and tractable model to study satiety sensing since the key components of physiological and metabolic regulations are largely conserved between mammals and flies. The research team led by WANG Liming has long channeled their time and energy into the neurobiological mechanisms of relevant behaviors, such as food intake and metabolism. In recent years, they have set up a relatively complete research paradigm to observe feeding behavior and metabolism in fruit flies and study the regulators of their feeding behavior. They have discovered that nutrients in food may not only alter the feeding behavior of fruit flies, but they may also alter their food-foraging behavior.
In this study, to identify neuropeptidergic cues that indicate the internal satiety state and suppress food consumption in turn, researchers carried out a neuron-specific RNAi screening in adult flies by using their previously developed MAFE (Manual Feeding) assay to examine food consumption. They ingeniously used non-nutritive L-glucose as a food source to circumvent the confounder of nutritive sugars to activate post-ingestive nutrient sensors and to modulate food consumption independent of satiety sensing.
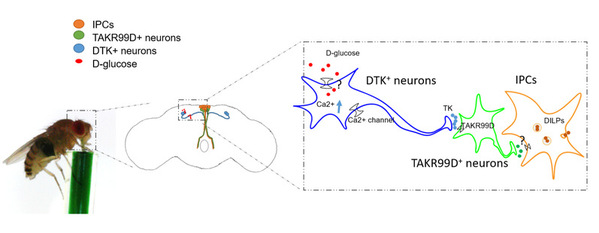
Researchers identified a neuropeptide receptor, tachykinin (DTK) receptor at 99D (TAKR99D), and its cognate ligand DTK, as potent feeding suppressors. This DTK–TAKR99D–IPC circuitry could be rapidly activated during food ingestion. Meanwhile, silencing and activating this neural circuitry exerted robust stimulatory and inhibitory effects on food consumption, respectively. Researchers thus hypothesized that DTK neurons function as a satiety sensor which can regulate feeding behavior in fruit flies.
To further test this hypothesis, researchers quantified DTK mRNA in starved and fed flies by using quantitative RT-PCR. In both head and body tissues, DTK expression was significantly reduced by starvation. They also found that re-feeding starved flies could rapidly induce DTK expression. Therefore, DTK–TAKR99D signaling might indeed be a satiety sensor.
The insulin-producing cells (IPCs) in the fly brain are one of the few well-studied regulators of feeding behavior and metabolism in fruit flies. IPCs can indirectly sense circulating glucose via other upstream neuronal populations including the adipokinetic hormone (AKH)-producing cells in the corpora cardiaca as well as a pair of glucose-sensing neurons in the fly brain expressing short neuropeptide F (sNPF) and corazonin (Crz). By means of such devices as GFP Reconstitution Across Synaptic Partners and optogenetics, researchers discovered that DTK+ neurons formed a two-synapse circuitry targeting insulin-producing cells, a well-known feeding suppressor, via TAKR99D+ neurons, and this circuitry could be rapidly activated during food ingestion and cease feeding.
Taken together, in this study, researchers identified a novel satiety sensor in the fly brain that could detect specific circulating nutrients and in turn modulate feeding, shedding light on satiety sensing mechanisms in mammals and how they are affected by metabolic disturbances.





