Scientists identify the structure of adhesion GPCR–G complexes
Recently, the research teams led by ZHANG Yan from the Zhejiang University School of Medicine and Zhejiang Laboratory for Systems & Precision Medicine, SUN Jinpeng from Peking University and Shandong University, and H. Eric Xu from the CAS Shanghai Institute of Materia Medica co-published an article entitled “Structures of glucocorticoid-bound adhesion receptor GPR97-Go complex” in Nature. It is the first time that the structure of adhesion GPCR–G complexes has been identified and analyzed.
Adhesion G-protein-coupled receptors (GPCRs) are a major family of GPCRs. As one of the evolutionarily ancient families in the GPCR superfamily, aGPCRs are crucial molecular switches that regulate many physiological processes, including brain development, ion–water homeostasis, inflammation and cell-fate determination. Mutations in aGPCRs have also been found to be associated with specific human diseases. Compared with other GPCR families, aGPCRs are featured by the presence of a large ectodomain that contains the GAIN domain, which functions together with the seven-transmembrane (7TM) bundle as a pair, and subsequent activation of the receptor through tethered agonism, mechanical force or other mechanisms. Although substantial progress has been made in discovering the emerging functions of aGPCRs, the coupling of several aGPCR members to G proteins remains to be clarified, the structural basis for aGPCR activation is unclear and whether the 7TM bundle of aGPCR constitutes a typical pocket to recognize a small chemical ligand is uncertain.
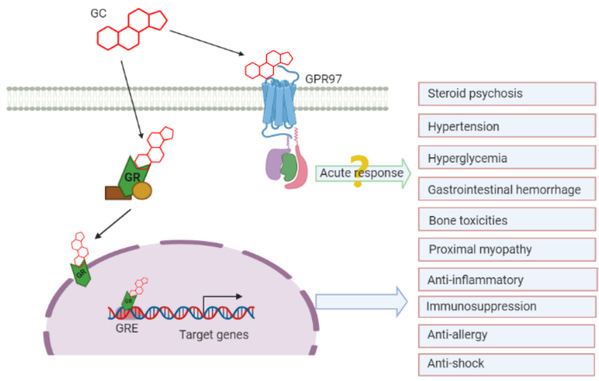
Glucocorticoids are part of the feedback mechanism in the immune system, which is very effective at reducing inflammation and suppressing the immune system. They are therefore widely used in medicine for inflammatory and autoimmune diseases. As early as 1950, Hench, Kendall and Reichstein were awarded with the Nobel Prize in Physiology and Medicine for the isolation and first therapeutic use of glucocorticoids. Previous studies indicate that glucocorticoids have two distinct modes of action. It is widely known that they bind to their nuclear receptors and function by mediating the expression of relevant genes in the nucleus. This mode, known as a genomic mechanism, often calls for a longer response time. In addition, researchers have found that glucocorticoids are also capable of triggering cellular and organismal changes via a rapid response. Further studies reveal that they can also bind to the membrane components of brain synapses. This suggests that there may well be membrane receptors. Follow-up studies demonstrate that the rapid response of glucocorticoids is closely related to G proteins and that PTX, an inhibitor of Gi, can inhibit the rapid action of glucocorticoids. Therefore, researchers have posited that the membrane receptor during the rapid response of glucocorticoids may well be GPCR. However, this speculation has yet to be confirmed and the mechanism of their rapid action remains enigmatic.
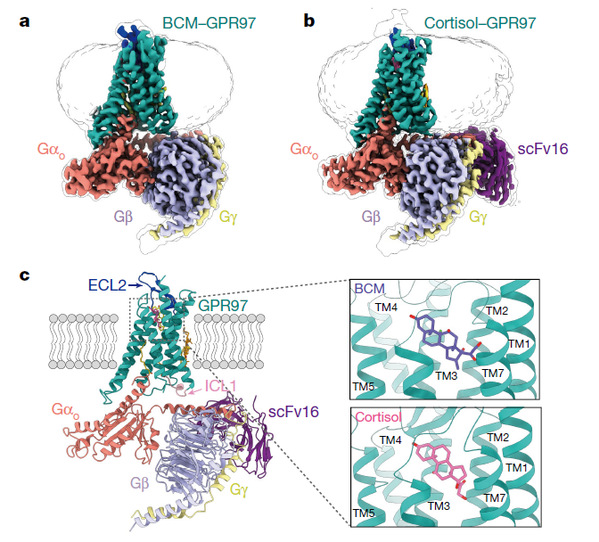
Previous studies show that the asthma drug beclomethasone propionate (BDP), a derivative of the anti-inflammatory drug beclomethasone (BCM) with two propionic acid modifications at positions 17 and 21, can activate GPR97 in vitro. On this basis, the team hypothesized that glucocorticoid analogs might be able to activate the GPR97 receptor. The team then screened endogenous steroid hormones systematically and eventually found that glucocorticoid hormones, including cortisol (hydrocortisone), cortisone and 11-deoxycortisol, can activate ligands for GPR97. Further experiments indicate that cortisol can regulate the Go signaling pathway by activating GPR97. Using a Titan Krios microscope, a total of 2,707 and 5,871 movies were collected for the BCM–GPR97-FL-AA–Go and the cortisol–GPR97-FL-AA–Go–scFv16 complexes reconstituted in vitro, respectively. The resulting cryo-EM maps after final refinement have overall resolutions of 3.1 Å and 2.9 Å.
The cryo-EM structure reveals that GPR97 has a unique 7TM architecture with an extended ECL2 β-sheet and a relatively short helix TM6. Although GPR97 has neither the traditional ‘PIF’ core triad nor the NPXXY motif that commonly mediates the activation of class A GPCRs, it senses ligand binding by the conserved toggle switch W6.53 and tethers TM3–TM5–TM6 by the newly identified and sequence-conserved upper Quaternary core for the active receptor conformation. In the aGPCR family, the conserved D/ERY motif is replaced by H(N)L(M)Y, which forms specific interactions with the palmitoylation of Go.
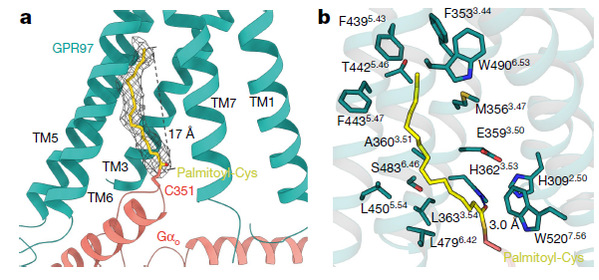
Remarkably, an unknown continuous electron density connected to C351 the α5 helix of Gαo was observed in the cortisol–GPR97–Go complex structure for the first time. The palmitoylation of C351 is universally acknowledged as a trigger for the loss of Gi/o signaling pathways in academia. However, this research reveals that palmitoylation at the cytosolic tail of the Go protein was found to be essential for efficient engagement with GPR97.
This work provides experimental evidence that endogenous steroid hormone glucocorticoids are high-affinity agonists for GPR97, an aGPCR, and presents the structures of GPR97–Go in complex with two glucocorticoids. It thus lays a structural basis for ligand binding to the seven-transmembrane domain of an adhesion GPCR and subsequent G protein coupling.





