Oncolytic virus therapy opens new horizons for advanced intrahepatic cholangiocarcinoma: FAHZU team publishes breakthrough study in Gut VG161 demonstrates promising immune-activating effects in advanced ICC
Oncolytic viral immunotherapy is rapidly emerging as a transformative strategy in solid tumor treatment. Now, a pioneering study by The First Affiliated Hospital, Zhejiang University School of Medicine (FAHZU) demonstrates—for the first time in humans—the potential of the multi-armored oncolytic virus VG161 to remodel the tumor microenvironment and bring clinical benefit to patients with advanced intrahepatic cholangiocarcinoma (ICC), one of the deadliest and most treatment-refractory liver cancers. The results were recently published in Gut.
ICC is among the most aggressive hepatobiliary malignancies, with global incidence rising sharply over recent decades. Most patients present at an advanced stage, when curative surgery is no longer possible. Conventional chemotherapy provides only limited survival improvement, and targeted therapies benefit only a small molecularly selected subgroup. Even immunotherapy—once considered a beacon of hope—often fails due to the profoundly immunosuppressive tumor microenvironment characteristic of ICC. This clinical gridlock calls for innovative therapeutic approaches capable of breaking tumor immune resistance.
The team led by Professor LIANG Tingbo, President of FAHZU and a global leader in hepatobiliary-pancreatic oncology innovation, previously developed the world-class third-generation oncolytic virus platform represented by VG161.
Building on years of translational immunovirology research, VG161 integrates: IL-12 to promote Th1 polarization, IL-15/IL-15Rα to support NK and CD8⁺ T-cell expansion, a PD-L1 blocking fusion protein to overcome checkpoint-mediated exhaustion, and a genetically optimized HSV-1 backbone for enhanced tumor selectivity. This multi-armed design enables VG161 to achieve both direct tumor oncolysis and multi-layered immune re-education.
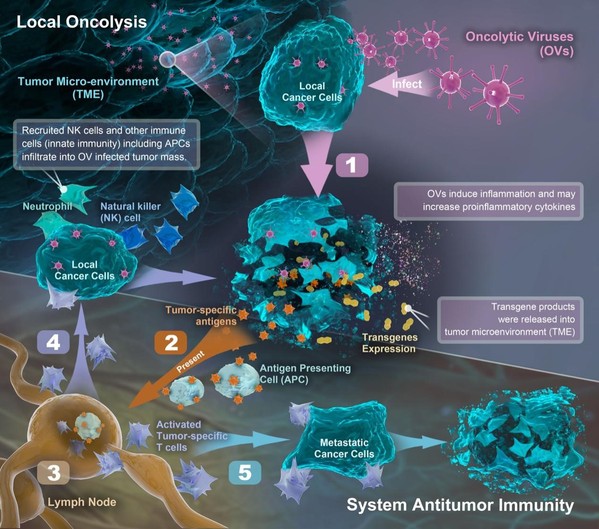
Figure 1. Schematic diagram expounding the mechanism of action of oncolytic virus
killing malignancies and enhancing tumor-specific immunity in TME
In the clinical study, twenty-four heavily pretreated ICC patients received ultrasound-guided intratumoral injections of VG161. Even in this late-line population, the results were encouraging: VG161 achieved a disease control rate of 31.8% and a median overall survival of 7.8 mFOX. Notably, patients with prior exposure to immune checkpoint inhibitors for more than three monthsonths, surpassing historical outcomes of the current second-line standard-of-care, FOL demonstrated markedly prolonged survival, reaching 10.3 months, suggesting that VG161 may reinvigorate exhausted immunity and potentially restore responsiveness to systemic immunotherapy.
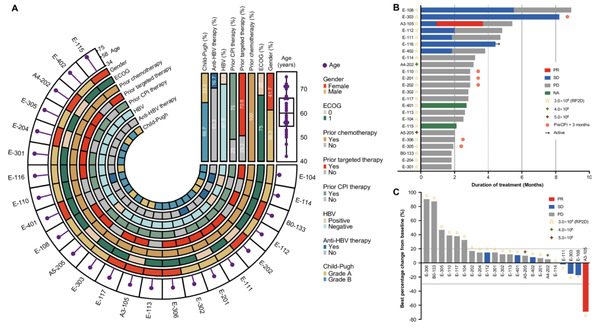
Figure 2. Clinical characteristics, treatment duration and response outcomes
To understand how VG161 exerts these effects, the team conducted an extensive multi-omics analysis using single-cell RNA sequencing, TCR sequencing, spatial immunofluorescence, and cytokine profiling. These analyses revealed profound immune reprogramming within the ICC microenvironment after treatment. CD8⁺ T cells and NK cells infiltrated the tumor in large numbers, accompanied by enhanced T-cell clonal expansion and activation of antigen-processing and interferon-response pathways. Both tumor-specific and virus-specific T-cell populations expanded simultaneously, with clinical responders exhibiting stronger activation signatures. The study also identified potential biomarkers of response, including a tumor epithelial subset associated with favorable outcomes and a macrophage population (Macro-C1QC) linked to immunosuppressive resistance. Spatial imaging confirmed that VG161 selectively localized within tumor tissue, further validating its tumor-targeting design.
These findings collectively show that VG161 is capable of converting ICC’s characteristically “cold” and suppressive tumor microenvironment into one that is immunologically active and responsive. Just as importantly, the improved outcomes observed in patients with prior immunotherapy exposure hint at the possibility of future treatment paradigms—such as a “sandwich” strategy—where VG161 reactivates antitumor immunity and enables subsequent systemic therapies to regain efficacy
“VG161 represents a powerful tool to overcome the immunological resistance of ICC,” said Professor LIANG Tingbo, corresponding author of the study. “Our findings provide strong rationale for integrating VG161 with checkpoint inhibitors in future clinical trials, with the hope of bringing more effective and durable treatment options to patients facing this devastating disease.”
Source: the First Affiliated Hospital, Zhejiang University School of Medicine (FAHZU)
Editor: HAN Xiao





