DAI Zhijun’s team develop a triple-targeting immunotherapy strategy to boost antitumor response through the CXCL16-CXCR6 axis
Cancer immunotherapy has transformed the treatment landscape for many malignancies, but a large proportion of patients still fail to achieve durable clinical benefit because tumors establish a profoundly immunosuppressive microenvironment. In particular, simultaneous immune evasion through impaired innate immune activation, TGF-β-driven suppression, and immune checkpoint signaling remains a major barrier to effective therapy.
Recently, DAI Zhijun’s team from Zhejiang University and collaborating institutions reported a new triple-targeting immunotherapy strategy that integrates STING activation with dual blockade of TGF-β and PD-L1. Their study, entitled “Triple targeting of STING, TGF-β, and PD-L1 boosts CXCL16-CXCR6 signaling for potent antitumor response,” was published in Nature Communications.
In this study, the researchers found that combining a STING agonist with the TGF-β×PD-L1 bispecific antibody YM101 achieved markedly stronger antitumor effects than conventional combinations in multiple preclinical tumor models. Mechanistically, they identified the CXCL16-CXCR6 axis as a key mediator of this synergy. STING activation together with TGF-β blockade promoted CXCL16 production in tumor-infiltrating myeloid cells, especially macrophages and dendritic cells, thereby recruiting and sustaining CXCR6+ cytotoxic CD8+ T cells within the tumor microenvironment. PD-L1 blockade further reinforced the antitumor activity of these T cells.
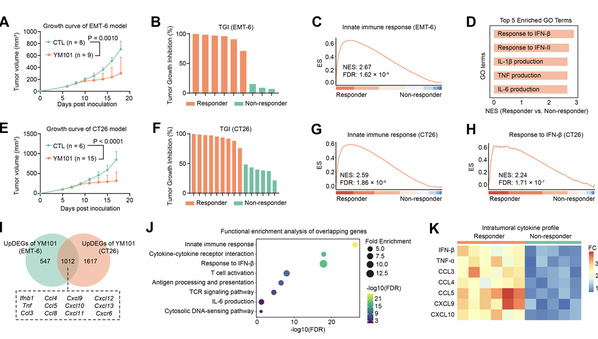
Figure 1. Distinct innate immune activation patterns are associated with differential response to YM101 treatment.
The team further demonstrated that this synergistic effect critically depends on the CXCL16-CXCR6 axis. Using multiple functional approaches, including neutralization experiments and genetically engineered mouse models, they showed that disruption of this pathway largely abolished the therapeutic benefit of the triple-targeting regimen. At the molecular level, they found that STING-driven type I interferon signaling activates STAT1, which directly promotes CXCL16 transcription, whereas TGF-β suppresses this process.
Based on these findings, the researchers developed Y101S, a novel immunostimulatory antibody conjugate that links a STING agonist payload to the TGF-β×PD-L1 bispecific antibody through a cleavable linker. In preclinical models, Y101S showed potent antitumor activity, enhanced immune memory, and improved intratumoral accumulation of CXCR6+CD8+ T cells, highlighting its translational potential for immunotherapy-resistant tumors.
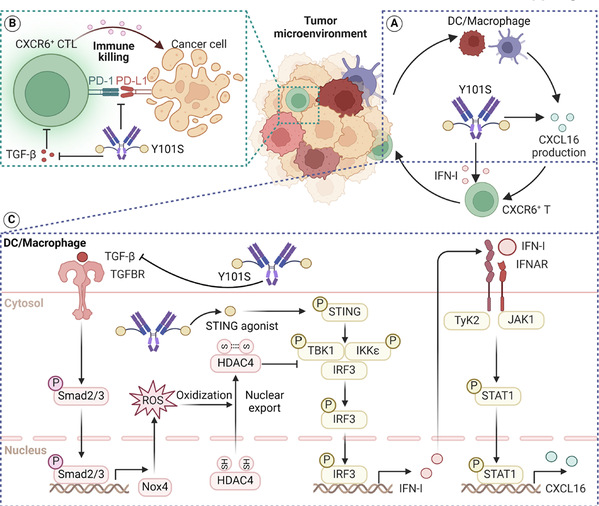
Figure 2. A schematic illustration of the triple-targeting strategy integrating STING activation with dual blockade of TGF-β and PD-L1 to enhance antitumor immunity.
This study provides a new framework for integrating innate immune activation with the relief of immune suppression and may open a promising avenue for treating resistant tumors,” said Professor DAI Zhijun, corresponding author of the study.
This work was led by researchers from The First Affiliated Hospital, Zhejiang University School of Medicine, in collaboration with colleagues from Tongji Hospital of Tongji Medical College, Huazhong University of Science and Technology, The Second Affiliated Hospital of Zhejiang University School of Medicine, Zhejiang University, Shanxi Bethune Hospital, and Wuhan YZY Biopharma Co., Ltd. YI Ming, LI Tianye, and GU Yinhui contributed equally to this work. The corresponding authors are YI Ming, CHU Qian, WU Kongming, and DAI Zhijun. This study was supported by the National Natural Science Foundation of China, the Natural Science Foundation of Zhejiang Province, and the Basic Research Project of Shanxi Science and Technology Department.
Source: The First Affiliated Hospital, Zhejiang University School of Medicine
Photo: The research team led by Prof. DAI Zhijun
Editor: HAN Xiao





