Iron-related cell death as a novel mechanism for heart diseases
A research team led by WANG Fudi, a professor at the School of Public Health, Zhejiang University, discovers that ferroptosis, a programmed iron-dependent cell death, as a mechanism in murine models of doxorubicin (DOX)- and ischemia/reperfusion (I/R)-induced cardiomyopathy. Five effective approaches are explored to preventing cardiac injuries, including ferroptosis inhibitor Fer-1, iron chelatordexrazoxane (DXZ), mitochondria-targeted antioxidant MitoTEMPO, Hmox1 inhibitor zinc protoporphyrin IX (ZnPP) and low-iron diets. This milestone discovery opens up a novel avenue for the prevention and treatment of cardiomyopathy. These findings are published in PNAS.
Heart disease, as one of the leading causes of death, needs to be cured in the field of well-being. A key pathogenic factor in the development of lethal heart failure is the loss of terminally differentiated cardiomyocytes. Therefore, it is of considerable research value and translational significance to mediate the death of cardiomyocytes. The death of cells can be broadly categorized into necrosis and apoptosis. With more in-depth research into the mechanisms for cell deaths, researchers have discovered a wide range of modes of cell death, such as necroptosis, autophagy, ferroptosis, pyroptosis and parthanatos. However, the type of cell death for cardiomyopathy is still open to dispute.

Ferroptosis is a novel form of regulated cell death characterized by the iron-dependent accumulation of lipid peroxides to lethal levels, which is morphologically, biochemically, and genetically distinct from apoptosis, necroptosis, and autophagy. Ferroptosis has been implicated in the pathological process associated with carcinogenesis, degenerative diseases, stroke, and kidney ischemia/reperfusion (I/R) injury.
To seek for a key regulator in the course of ferroptosis in models of DOX-induced cardiomyopathy, WANG Fudi et al. discover thatheme oxygenase-1 (Hmox1) is significantly up-regulated in DOX-treated murine hearts by means of RNA-sequencing. Administering DOX to mice induces cardiomyopathy with a rapid, systemic accumulation of nonheme iron via heme degradation by Nrf2-mediated up-regulation of Hmox1, whose effect isabsent in Nrf2-deficent mice. Conversely, zinc protoporphyrin IX, an Hmox1 antagonist, protects the DOX-treated mice, suggesting free iron released on heme degradation is essential and sufficient to induce cardiac injuries.
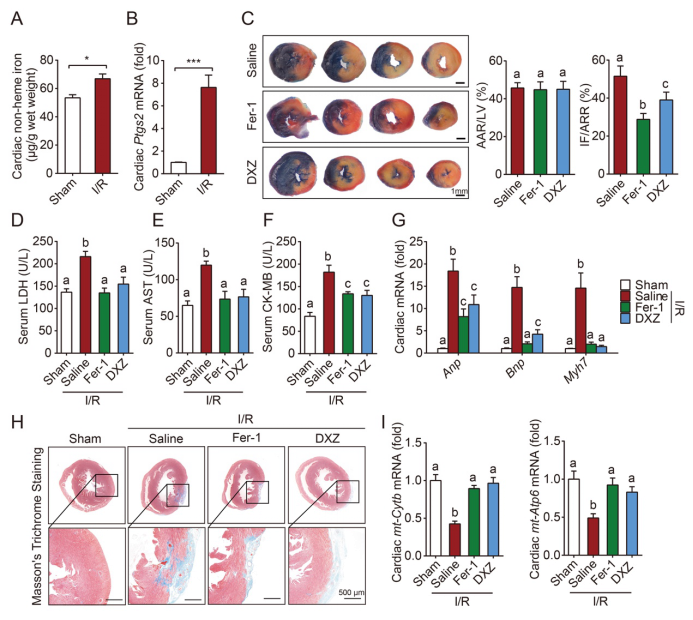
Given that ferroptosis is driven by damage to lipid membranes, WANG Fudi et al. imply that excess free iron is accumulated in mitochondria and causes lipid peroxidation on its membrane. MitoTEMPO significantly rescues DOX cardiomyopathy, supporting oxidative damage to mitochondria as a major mechanism in ferroptosis-induced heart damage. Importantly, ferrostatin-1 and iron chelation also ameliorate heart failure induced by both acute and chronic I/R in mice. These findings highlight that targeting ferroptosis serves as a promising way to prevent cardiomyopathy effectively.





